Legacy of Repeated Cultivation Drives Cyclical Microbial Community Development in a Tropical Oxisol Soil
- PMID: 40240613
- PMCID: PMC12003493
- DOI: 10.1007/s00248-025-02530-3
Legacy of Repeated Cultivation Drives Cyclical Microbial Community Development in a Tropical Oxisol Soil
Abstract
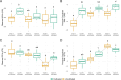
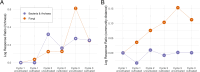
Agricultural practices and the crop being actively cultivated are some of the most important contributors to soil microbial community assembly processes in agroecosystems. However, it is not well-understood how the cultivation of diverse crop species can directionally shift complex soil microbial communities, especially under continuous monoculture systems. Here, we conducted a field experiment to assess how three crop species (Lactuca sativa, Brassica juncea, and Zea mays) may shift soil microbial (bacteria/archaea and fungi) communities when planted in a monoculture and repeatedly grown for three cycles in a tropical Oxisol soil. We found that while plant species made limited contributions to microbial community differentiation, repeated cultivation was a strong driver of community development over time. The bacterial/archaeal communities exhibited a cyclical community development pattern, initially with strong differentiation that attenuated to a steady state at the end of the three cycles. In contrast, fungal communities generally developed more linearly and may have only started to stabilize after three cropping cycles. These developments may speak to the stronger legacy effects on fungal communities. Together, these results highlight the differences between how bacteria/archaea and fungal communities develop, especially in tropical, underdeveloped, intensively degraded, or marginal soils.
Keywords: Agroecosystems; Crop microbiome; Legacy effect; Monoculture; Temporal community development.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing Interests: The authors declare no competing interests.
Figures


Similar articles
-
Soil microbial community composition by crop type under rotation diversification.BMC Microbiol. 2024 Oct 26;24(1):435. doi: 10.1186/s12866-024-03580-2. BMC Microbiol. 2024. PMID: 39462325 Free PMC article.
-
Maize edible-legumes intercropping systems for enhancing agrobiodiversity and belowground ecosystem services.Sci Rep. 2024 Jun 21;14(1):14355. doi: 10.1038/s41598-024-64138-w. Sci Rep. 2024. PMID: 38906908 Free PMC article.
-
Soil prokaryotic and fungal biome structures associated with crop disease status across the Japan Archipelago.mSphere. 2024 Apr 23;9(4):e0080323. doi: 10.1128/msphere.00803-23. Epub 2024 Apr 3. mSphere. 2024. PMID: 38567970 Free PMC article.
-
Metagenomic profiling of rhizosphere microbial community structure and diversity associated with maize plant as affected by cropping systems.Int Microbiol. 2021 Aug;24(3):325-335. doi: 10.1007/s10123-021-00169-x. Epub 2021 Mar 5. Int Microbiol. 2021. PMID: 33666787
-
Dynamic changes of rhizosphere soil bacterial community and nutrients in cadmium polluted soils with soybean-corn intercropping.BMC Microbiol. 2022 Feb 15;22(1):57. doi: 10.1186/s12866-022-02468-3. BMC Microbiol. 2022. PMID: 35168566 Free PMC article.
References
-
- Brunel C, Pouteau R, Dawson W et al (2020) Towards unraveling macroecological patterns in rhizosphere microbiomes. Trends Plant Sci. 10.1016/j.tplants.2020.04.015 - PubMed
-
- Yang T, Lupwayi N, Marc S-A et al (2021) Anthropogenic drivers of soil microbial communities and impacts on soil biological functions in agroecosystems. Glob Ecol Conserv 27:e01521–e01521. 10.1016/j.gecco.2021.e01521
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

