Potentiating doxorubicin activity through BCL-2 inhibition in p53 wild-type and mutated triple-negative breast cancer
- PMID: 40242242
- PMCID: PMC11999952
- DOI: 10.3389/fonc.2025.1549282
Potentiating doxorubicin activity through BCL-2 inhibition in p53 wild-type and mutated triple-negative breast cancer
Abstract
Background: Triple-negative breast cancer (TNBC) is an aggressive sub-type of breast cancer that is associated with higher rates of recurrent disease. Chemotherapy with an anthracycline is an integral part of curative therapy but resistance remains a clinical problem. Cellular senescence is a terminal cell fate that has been observed in models of doxorubicin resistance. Identifying novel combinations with doxorubicin to eliminate senescent cells and promote apoptosis may lead to improved clinical outcomes. The purpose of this study was to investigate the combination of doxorubicin with the pro-apoptotic BCL-2 inhibitor venetoclax in TNBC cell lines and to assess the role of p53 in cellular senescence and apoptosis.
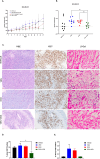
Methods: TNBC cell lines with wild-type (WT), mutated or knocked-down (KD) p53 were treated with doxorubicin, venetoclax or the combination in vitro and evaluated for impacts on viability, proliferation, apoptosis, and senescence. Down-stream markers of apoptosis were also assessed to evaluate cellular mechanistic changes. An in vivo TNBC MDA-MB-231 murine model was used to assess tumor growth, cellular proliferation, and senescence changes following treatment with doxorubicin, venetoclax or combination.
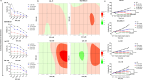
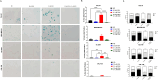
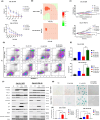
Results: Venetoclax with doxorubicin had synergistic antiproliferative activity against TNBC cell lines and increased apoptosis. The addition of venetoclax to doxorubicin reduced senescent cells in a p53-independent manner. In vivo, the addition of venetoclax to doxorubicin improved tumor growth inhibition and reduced senescent cells.
Conclusion: The combination of doxorubicin with venetoclax is promising for the treatment of p53-WT and mutated TNBC and this work supports further investigation.
Keywords: TNBC; doxorubicin; resistance; senescence; venetoclax.
Copyright © 2025 Schreiber, Smoots, Jackson, Bagby, Dus, Dominguez, Binns, Pitts and Diamond.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures


Similar articles
-
Overcoming doxorubicin resistance in triple-negative breast cancer using the class I-targeting HDAC inhibitor bocodepsin/OKI-179 to promote apoptosis.Breast Cancer Res. 2024 Mar 1;26(1):35. doi: 10.1186/s13058-024-01799-5. Breast Cancer Res. 2024. PMID: 38429789 Free PMC article.
-
Synergistic Anti-Tumor Effects of Newcastle Disease Virus and Doxorubicin: Evidence from A Murine Breast Cancer Model.Int Immunopharmacol. 2024 Dec 25;143(Pt 2):113481. doi: 10.1016/j.intimp.2024.113481. Epub 2024 Oct 29. Int Immunopharmacol. 2024. PMID: 39467343
-
Efficacy and Molecular Mechanisms of Differentiated Response to the Aurora and Angiogenic Kinase Inhibitor ENMD-2076 in Preclinical Models of p53-Mutated Triple-Negative Breast Cancer.Front Oncol. 2017 May 15;7:94. doi: 10.3389/fonc.2017.00094. eCollection 2017. Front Oncol. 2017. PMID: 28555173 Free PMC article.
-
Epigenetic Co-Deregulation of EZH2/TET1 is a Senescence-Countering, Actionable Vulnerability in Triple-Negative Breast Cancer.Theranostics. 2019 Jan 24;9(3):761-777. doi: 10.7150/thno.29520. eCollection 2019. Theranostics. 2019. PMID: 30809307 Free PMC article.
-
p53 in breast cancer subtypes and new insights into response to chemotherapy.Breast. 2013 Aug;22 Suppl 2:S27-9. doi: 10.1016/j.breast.2013.07.005. Breast. 2013. PMID: 24074787 Review.
Cited by
-
Cellular senescence in cancer: from mechanism paradoxes to precision therapeutics.Mol Cancer. 2025 Aug 8;24(1):213. doi: 10.1186/s12943-025-02419-2. Mol Cancer. 2025. PMID: 40781676 Free PMC article. Review.
References
-
- Lin NU, Vanderplas A, Hughes ME, Theriault RL, Edge SB, Wong YN, et al. . Clinicopathologic features, patterns of recurrence, and survival among women with triple-negative breast cancer in the National Comprehensive Cancer Network. Cancer. (2012) 118:5463–72. doi: 10.1002/cncr.v118.22 - DOI - PMC - PubMed
-
- National Comprehensive Cancer Network . NCCN clinical practice guidelines in oncology: breast cancer Version 4.2023 (2023). Available online at: https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf (Accessed March 28, 2024).
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

