Supramolecular Recognition of a DNA Four-Way Junction by an M2L4 Metallo-Cage, Inspired by a Simulation-Guided Design Approach
- PMID: 40243103
- PMCID: PMC12184311
- DOI: 10.1002/anie.202504866
Supramolecular Recognition of a DNA Four-Way Junction by an M2L4 Metallo-Cage, Inspired by a Simulation-Guided Design Approach
Abstract
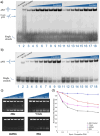
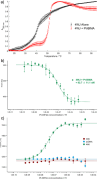
DNA four-way junctions (4WJs) play an important biological role in DNA repair and recombination, and viral regulation, and are attractive therapeutic targets. Compounds that recognise the junction structure are rare; in this work, we describe cationic metallo-supramolecular M2L4 cages as a new type of 4WJ binder with nanomolar affinities. A combination of molecular dynamics (MD) simulations and biophysical experiments show that the size and shape of a compound comprising square planar Pd or Pt and anthracene-based ligands is an excellent fit for the 4WJ cavity. Whilst the cage is also capable of binding to three-way junctions (3WJs) and Y-fork structures, we show that the 4WJ is the preferred DNA target, and that duplex B-DNA is not a competitor. Among 3WJs, T-shape bulged 3WJs are bound more preferably than perfect Y-shaped 3WJs. Whilst previous work studying M2L4 metallo-supramolecular cages has focused on binding inside their structures, this work exploits the external aromatic surfaces of the supramolecule, creating a supramolecular guest that ideally matches the DNA host cavity. This approach allows available structures to be identified as potential junction binders and then screened for their fit to a nucleic acid junction target using simulations. This has potential to significantly accelerate discovery.
Keywords: Bio‐inorganic; DNA four‐way junctions; DNA‐recognition; Metallo‐cage; Supramolecular chemistry.
© 2025 The Author(s). Angewandte Chemie International Edition published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Kenny R. G., Marmion C. J., Chem. Rev. 2019, 119, 1058–1137. - PubMed
-
- Rottenberg S., Disler C., Perego P., Nat. Rev. Cancer 2021, 21, 37–50. - PubMed
-
- Martins‐Teixeira M. B., Carvalho I., ChemMedChemChemMedChem 2020, 15, 933–948. - PubMed
-
- Ivens E., Cominetti M. M. D., Searcey M., Bioorg. Med. Chem. 2022, 69, 116897. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

