Carnitine Deficiency Caused by Salcaprozic Acid Sodium Contained in Oral Semaglutide in a Patient with Multiple Acyl-CoA Dehydrogenase Deficiency
- PMID: 40243535
- PMCID: PMC11989126
- DOI: 10.3390/ijms26072962
Carnitine Deficiency Caused by Salcaprozic Acid Sodium Contained in Oral Semaglutide in a Patient with Multiple Acyl-CoA Dehydrogenase Deficiency
Abstract
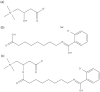
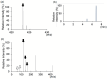
Carnitine plays an essential role in maintaining energy homeostasis and metabolic flexibility. Various medications, such as pivalate-conjugated antibiotics, valproic acid, and anticancer agents, can induce carnitine deficiency, inhibit the utilization of fatty acid, and contribute to the development of hypoglycemia. No studies have linked oral semaglutide to carnitine deficiency. Herein, we report the case of a 34-year-old male patient with multiple acyl-CoA dehydrogenase deficiency who developed carnitine deficiency attributable to salcaprozic acid sodium (SNAC) in oral semaglutide. The patient was diagnosed with type 2 diabetes mellitus at 32 years of age and was treated with semaglutide injections. Hypoglycemic symptoms appeared after switching to oral semaglutide, and the mean levels of blood-free carnitine significantly decreased. Liquid chromatography-tandem mass spectrometry analysis revealed a peak corresponding to the SNAC-carnitine complex (m/z 423.24) in the urine exclusively during the oral administration of semaglutide. The MS/MS spectra at m/z 423.24 contained peaks consistent with those of the SNAC and carnitine product ions. Our results suggest that through complexation with carnitine, SNAC may induce carnitine deficiency. Healthcare providers should monitor for carnitine deficiency when administering SNAC-containing medications to at-risk individuals. Furthermore, this case can raise more significant concerns about the potential impact of pharmaceutical excipients like SNAC on metabolic pathways.
Keywords: carnitine deficiency; multiple acyl-CoA dehydrogenase deficiency; oral semaglutide; salcaprozic acid sodium; type 2 diabetes.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Safety and Pharmacokinetics of Single and Multiple Ascending Doses of the Novel Oral Human GLP-1 Analogue, Oral Semaglutide, in Healthy Subjects and Subjects with Type 2 Diabetes.Clin Pharmacokinet. 2019 Jun;58(6):781-791. doi: 10.1007/s40262-018-0728-4. Clin Pharmacokinet. 2019. PMID: 30565096 Clinical Trial.
-
Effect of Oral Semaglutide on the Pharmacokinetics of Lisinopril, Warfarin, Digoxin, and Metformin in Healthy Subjects.Clin Pharmacokinet. 2019 Sep;58(9):1193-1203. doi: 10.1007/s40262-019-00756-2. Clin Pharmacokinet. 2019. PMID: 30945118 Free PMC article. Clinical Trial.
-
A new era for oral peptides: SNAC and the development of oral semaglutide for the treatment of type 2 diabetes.Rev Endocr Metab Disord. 2022 Oct;23(5):979-994. doi: 10.1007/s11154-022-09735-8. Epub 2022 Jul 15. Rev Endocr Metab Disord. 2022. PMID: 35838946 Free PMC article. Review.
-
Oral Semaglutide: A Review of the First Oral Glucagon-Like Peptide 1 Receptor Agonist.Diabetes Technol Ther. 2020 Jan;22(1):10-18. doi: 10.1089/dia.2019.0185. Epub 2019 Oct 1. Diabetes Technol Ther. 2020. PMID: 31436480 Review.
-
Follow-up of fatty acid β-oxidation disorders in expanded newborn screening era.Eur J Pediatr. 2019 Mar;178(3):387-394. doi: 10.1007/s00431-018-03315-2. Epub 2019 Jan 7. Eur J Pediatr. 2019. PMID: 30617651
References
-
- Carter A.L., Abney T.O., Lapp D.F. Biosynthesis and Metabolism of Carnitine. J. Child Neurol. 1995;10:S3–S7. - PubMed
-
- McGarry J.D., Brown N.F. The Mitochondrial Carnitine Palmitoyltransferase System. From Concept to Molecular Analysis. Eur. J. Biochem. 1997;244:1–14. - PubMed
-
- Ramsay R.R., Zammit V.A. Carnitine Acyltransferases and Their Influence on CoA Pools in Health and Disease. Mol. Asp. Med. 2004;25:475–493. - PubMed
-
- Pons R., De Vivo D.C. Primary and Secondary Carnitine Deficiency Syndromes. J. Child Neurol. 1995;10:S8–S24. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

