From DNA Repair to Redox Signaling: The Multifaceted Role of APEX1 (Apurinic/Apyrimidinic Endonuclease 1) in Cardiovascular Health and Disease
- PMID: 40243693
- PMCID: PMC11988304
- DOI: 10.3390/ijms26073034
From DNA Repair to Redox Signaling: The Multifaceted Role of APEX1 (Apurinic/Apyrimidinic Endonuclease 1) in Cardiovascular Health and Disease
Abstract
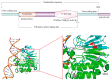
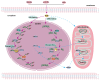
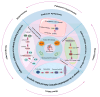
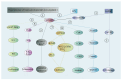
Apurinic/apyrimidinic endonuclease 1 (APEX1) serves as a potent regulatory factor in innate immunity, exhibiting both redox and endonuclease activities. Its redox function enables the regulation of transcription factors such as NF-κB or STAT3, whereas its endonuclease activity recognizes apurinic/apyrimidinic (AP) sites in damaged DNA lesions during base excision repair (BER) and double-stranded DNA repair, thereby I confirm.anti-inflammatory, antioxidative stress and antiapoptotic effects. APEX1 is expressed in a variety of cell types that constitute the cardiovascular system, including cardiomyocytes, endothelial cells, smooth muscle cells, and immune cells. Emerging genetic and experimental evidence points towards the functional roles of APEX1 in the pathophysiology of cardiovascular diseases, including neointimal formation and atherosclerosis. This review aims to present comprehensive coverage of the up-to-date literature concerning the molecular and cellular functions of APEX1, with a particular focus on how APEX1 contributes to the (dys)functions of different cell types during the pathogenesis of cardiovascular diseases. Furthermore, we underscore the potential of APEX1 as a therapeutic target for the treatment of cardiovascular diseases.
Keywords: APEX1; DNA repair; atherosclerosis; cardiovascular diseases; endonuclease activity; redox activity.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Mild phenotype of knockouts of the major apurinic/apyrimidinic endonuclease APEX1 in a non-cancer human cell line.PLoS One. 2021 Sep 16;16(9):e0257473. doi: 10.1371/journal.pone.0257473. eCollection 2021. PLoS One. 2021. PMID: 34529719 Free PMC article.
-
The role of the N-terminal domain of human apurinic/apyrimidinic endonuclease 1, APE1, in DNA glycosylase stimulation.DNA Repair (Amst). 2018 Apr;64:10-25. doi: 10.1016/j.dnarep.2018.02.001. Epub 2018 Feb 11. DNA Repair (Amst). 2018. PMID: 29475157
-
Endonuclease and redox activities of human apurinic/apyrimidinic endonuclease 1 have distinctive and essential functions in IgA class switch recombination.J Biol Chem. 2019 Mar 29;294(13):5198-5207. doi: 10.1074/jbc.RA118.006601. Epub 2019 Jan 31. J Biol Chem. 2019. PMID: 30705092 Free PMC article.
-
Inhibitors of nuclease and redox activity of apurinic/apyrimidinic endonuclease 1/redox effector factor 1 (APE1/Ref-1).Bioorg Med Chem. 2017 May 1;25(9):2531-2544. doi: 10.1016/j.bmc.2017.01.028. Epub 2017 Jan 21. Bioorg Med Chem. 2017. PMID: 28161249 Review.
-
AP Endonuclease 1 as a Key Enzyme in Repair of Apurinic/Apyrimidinic Sites.Biochemistry (Mosc). 2016 Sep;81(9):951-67. doi: 10.1134/S0006297916090042. Biochemistry (Mosc). 2016. PMID: 27682167 Review.
References
-
- Demple B., Linn S. On the recognition and cleavage mechanism of Escherichia coli endodeoxyribonuclease V, a possible DNA repair enzyme. J. Biol. Chem. 1982;257:2848–2855. - PubMed
-
- Linsley W.S., Penhoet E.E., Linn S. Human endonuclease specific for apurinic/apyrimidinic sites in DNA. Partial purification and characterization of mul-tiple forms from placenta. J. Biol. Chem. 1977;252:1235–1242. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

