Can Humanized Immune System Mouse and Rat Models Accelerate the Development of Cytomegalovirus-Based Vaccines Against Infectious Diseases and Cancers?
- PMID: 40243710
- PMCID: PMC11988357
- DOI: 10.3390/ijms26073082
Can Humanized Immune System Mouse and Rat Models Accelerate the Development of Cytomegalovirus-Based Vaccines Against Infectious Diseases and Cancers?
Abstract
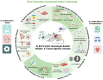
Over the past three decades, immunodeficient mouse models carrying human immune cells, with or without human lymphoid tissues, termed humanized immune system (HIS) rodent models, have been developed to recapitulate the human immune system and associated immune responses. HIS mouse models have successfully modeled many human-restricted viral infections, including those caused by human cytomegalovirus (HCMV) and human immunodeficiency virus (HIV). HIS mouse models have also been used to model human cancer immunobiology, which exhibits differences from murine cancers in traditional mouse models. Variants of HIS mouse models that carry human liver cells, lung tissue, skin tissue, or human patient-derived tumor xenografts and human hematopoietic stem cells-derived-human immune cells with or without lymphoid tissue xenografts have been developed to probe human immune responses to infections and human tumors. HCMV-based vaccines are human-restricted, which poses limitations for mechanistic and efficacy studies using traditional animal models. The HCMV-based vaccine approach is a promising vaccine strategy as it induces robust effector memory T cell responses that may be critical in preventing and rapidly controlling persistent viral infections and cancers. Here, we review novel HIS mouse models with robust human immune cell development and primary and secondary lymphoid tissues that could address many of the limitations of HIS mice in their use as animal models for HCMV-based vaccine research. We also reviewed novel HIS rat models, which could allow long-term (greater than one year) vaccinology studies and better recapitulate human pathophysiology. Translating laboratory research findings to clinical application is a significant bottleneck in vaccine development; HIS rodents and related variants that more accurately model human immunology and diseases could increase the translatability of research findings.
Keywords: HCMV-based vaccines; HIV vaccines; HIV/AIDS-animal models; human cancer xenograft models; humanized immune system and rats.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Agarwal Y., Beatty C., Biradar S., Castronova I., Ho S., Melody K., Bility M.T. Moving beyond the mousetrap: Current and emerging humanized mouse and rat models for investigating prevention and cure strategies against HIV infection and associated pathologies. Retrovirology. 2020;17:8. doi: 10.1186/s12977-020-00515-3. - DOI - PMC - PubMed
-
- Akkina R., Barber D.L., Bility M.T., Bissig K.-D., Burwitz B.J., Eichelberg K., Endsley J.J., Garcia J.V., Hafner R., Karakousis P.C., et al. Small Animal Models for Human Immunodeficiency Virus (HIV), Hepatitis B, and Tuberculosis: Proceedings of an NIAID Workshop. Curr. HIV Res. 2020;18:19–28. doi: 10.2174/1570162X18666191223114019. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

