Unlocking the Complexity of Antibody-Drug Conjugates: A Cutting-Edge LC-HRMS Approach to Refine Drug-to-Antibody Ratio Measurements with Highly Reactive Payloads
- PMID: 40243720
- PMCID: PMC11988793
- DOI: 10.3390/ijms26073080
Unlocking the Complexity of Antibody-Drug Conjugates: A Cutting-Edge LC-HRMS Approach to Refine Drug-to-Antibody Ratio Measurements with Highly Reactive Payloads
Abstract
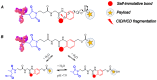
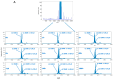
The complexity of therapeutic proteins like antibody-drug conjugates (ADCs) holds a tremendous analytical challenge. Complementary mass spectrometry approaches such as peptide mapping and intact mass analysis are required for the in-depth characterization of these bioconjugates. Cysteine-linked ADCs have shown a unique challenge for characterization, mainly when the conjugation is carried out on interchain cysteines, because their intact analysis requires native mass spectrometry conditions to preserve non-covalent binding between antibody chains. In this work, two different approaches were proposed. Specifically, a full scan data-independent all ion fragmentation (FS-AIF) and a full scan data-dependent targeted MS2 (FS-ddtMS2) were applied to generate complementary datasets for a cysteine-linked ADC characterization with a highly reactive payload. These two methods were applied to in vitro plasma stability and in vivo PK samples to calculate and refine mean drug-to-antibody ratio over time. Using this approach, we successfully characterized an ADC containing a hydrolysis-sensitive payload and refined the "active" drug-to-antibody ratio on in vitro stability and in vivo samples. These two methods allowed the confirmation of the different ADC species and potential metabolites of conjugated payload attached to the antibody backbone in a single analysis without needing a dedicated method for the conjugated payload metabolite identification.
Keywords: antibody–drug conjugates; drug discovery; drug-to-antibody ratio; liquid-chromatography mass spectrometry.
Conflict of interest statement
KJC, LB, and FRS were employed by the company Merck KGaA. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures






References
-
- Abdollahpour-Alitappeh M., Lotfinia M., Gharibi T., Mardaneh J., Farhadihosseinabadi B., Larki P., Faghfourian B., Sepehr K.S., Abbaszadeh-Goudarzi K., Abbaszadeh-Goudarzi G., et al. Antibody–drug conjugates (ADCs) for cancer therapy: Strategies, challenges, and successes. J. Cell. Physiol. 2019;234:5628–5642. doi: 10.1002/jcp.27419. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

