Pathological Changes in Extracellular Matrix Composition Orchestrate the Fibrotic Feedback Loop Through Macrophage Activation in Dupuytren's Contracture
- PMID: 40243889
- PMCID: PMC11988646
- DOI: 10.3390/ijms26073146
Pathological Changes in Extracellular Matrix Composition Orchestrate the Fibrotic Feedback Loop Through Macrophage Activation in Dupuytren's Contracture
Abstract
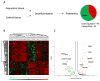
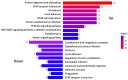
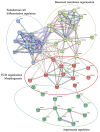
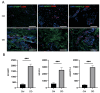
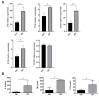
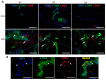
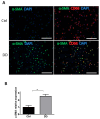
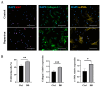
Dupuytren's contracture belongs to a group of fibrotic diseases that have similar mechanisms but lack effective treatment and prevention options. The excessive accumulation of connective tissue in Dupuytren's disease leads to palmar fibrosis that results in contracture deformities. The present study aimed to investigate how the tissue microenvironment in Dupuytren's contracture affects the phenotypic differentiation of macrophages, which leads to an inflammatory response and the development of chronicity in fibrotic disease. We utilized a decellularization-based method combined with proteomic analysis to identify shifts in extracellular matrix composition and the surrounding tissue microenvironment. We found that the expression of several matricellular proteins, such as MFAP4, EFEMP1 (fibulin-3), and ANGPTL2, was elevated in Dupuytren's tissue. We show that, in response to the changes in the extracellular matrix of Dupuytren's contracture, macrophages regulate the fibrotic process by cytokine production, promote myofibroblast differentiation, and increase the fibroblast migration rate. Moreover, we found that the extracellular matrix of Dupuytren's contracture directly supports the macrophage-to-myofibroblast transition, which could be another contributor to Dupuytren's disease pathogenesis. Our results suggest that interactions between macrophages and the extracellular matrix should be considered as targets for novel fibrotic disease treatment and prevention strategies in the future.
Keywords: Dupuytren’s contracture; extracellular matrix; fibrosis; macrophages; myofibroblasts.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Unraveling the signaling pathways promoting fibrosis in Dupuytren's disease reveals TNF as a therapeutic target.Proc Natl Acad Sci U S A. 2013 Mar 5;110(10):E928-37. doi: 10.1073/pnas.1301100110. Epub 2013 Feb 19. Proc Natl Acad Sci U S A. 2013. PMID: 23431165 Free PMC article.
-
Involvement of pro-inflammatory cytokines and growth factors in the pathogenesis of Dupuytren's contracture: a novel target for a possible future therapeutic strategy?Clin Sci (Lond). 2015 Oct 1;129(8):711-20. doi: 10.1042/CS20150088. Epub 2015 Jun 11. Clin Sci (Lond). 2015. PMID: 26201022
-
Identification of histological patterns in clinically affected and unaffected palm regions in dupuytren's disease.PLoS One. 2014 Nov 7;9(11):e112457. doi: 10.1371/journal.pone.0112457. eCollection 2014. PLoS One. 2014. PMID: 25379672 Free PMC article.
-
[The pathobiochemistry of Dupuytren's contracture].Chir Narzadow Ruchu Ortop Pol. 2009 Mar-Apr;74(2):68-73. Chir Narzadow Ruchu Ortop Pol. 2009. PMID: 19514484 Review. Polish.
-
[Pathogenesis of Dupuytren's contracture--a review].Chir Narzadow Ruchu Ortop Pol. 2002;67(1):73-9. Chir Narzadow Ruchu Ortop Pol. 2002. PMID: 12087679 Review. Polish.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

