Hydroxyurea Mitigates Heme-Induced Inflammation and Kidney Injury in Humanized Sickle Cell Mice
- PMID: 40244015
- PMCID: PMC11989777
- DOI: 10.3390/ijms26073214
Hydroxyurea Mitigates Heme-Induced Inflammation and Kidney Injury in Humanized Sickle Cell Mice
Abstract
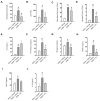
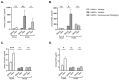
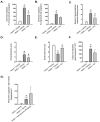
Kidney disorders significantly contribute to morbidity and mortality in sickle cell disease (SCD). Acute kidney injury (AKI), a major risk factor for chronic kidney disease (CKD), often arises from intravascular hemolysis, where plasma cell-free heme drives AKI through inflammatory and oxidative stress mechanisms. Hydroxyurea (HU), a well-established SCD-modifying therapy, improves clinical outcomes, but its effects on systemic heme and inflammatory mediators of kidney injury remain underexplored. This study evaluated HU's impact on plasma heme, pro-inflammatory mediators, kidney injury, and renal histopathology in a sickle cell mouse model. Townes humanized sickle cell mice (HbSS) and non-sickle (HbAA) controls were treated with HU or vehicle for two weeks. HU significantly reduced total plasma heme, lactate dehydrogenase, and pro-inflammatory cytokines (CXCL10, VEGF-A, IFN-γ) in HbSS mice. HU reduced renal injury biomarkers (cystatin C, NGAL) and improved renal histopathology, evidenced by reduced vascular congestion, glomerulosclerosis, and tubular damage. Interestingly, HU did not alter the levels of kidney repair biomarkers (clusterin and EGF). These findings suggest that HU mitigates kidney injury by reducing the deleterious effects of circulating heme and inflammation, supporting its potential to slow or prevent progressive kidney injury in SCD.
Keywords: acute kidney injury; heme; hydroxyurea; inflammation; sickle cell disease.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Thomson A.M., McHugh T.A., Oron A.P., Teply C., Lonberg N., Vilchis Tella V., Wilner L.B., Fuller K., Hagins H., Aboagye R.G., et al. Global, Regional, and National Prevalence and Mortality Burden of Sickle Cell Disease, 2000–2021: A Systematic Analysis from the Global Burden of Disease Study 2021. Lancet Haematol. 2023;10:e585–e599. doi: 10.1016/S2352-3026(23)00118-7. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

