Comprehensive analysis of species-specific differences in fatty acid composition and proteome of milk fat globules in human and animals
- PMID: 40248317
- PMCID: PMC12005228
- DOI: 10.1016/j.fochx.2025.102431
Comprehensive analysis of species-specific differences in fatty acid composition and proteome of milk fat globules in human and animals
Abstract
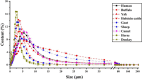
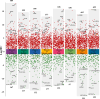
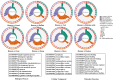
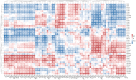
Human milk, the gold standard for infant nutrition, precisely captures infant needs. This study analyzed the particle size distribution, fatty acid (FA) profile, and milk fat globule membrane (MFGM) proteome of milk fat globules (MFGs) in human milk and eight types of animal milks. Donkey and horse milks showed a greater abundance of smaller MFGs (0-5 μm). Human milk was richer in monounsaturated FAs, such as oleic acid and nervonic acid, whereas donkey had the most polyunsaturated FAs like linoleic acid and cis-11,14-eicosadienoic acid. Among the identified 1253 MFGM proteins, human milk showed significantly higher abundance of several novel immune enhancers including adiponectin B and vitronectin, antioxidases, and lipid metabolism regulators. Meanwhile, sheep milk and yak milk displayed greater abundance of zinc-α-2-glycoprotein and selenoprotein F, respectively. This study revealed physicochemical differences in MFGs between humans and eight types of animals, offering insights for improving animal-milk-based formulas for infants' nutritional needs.
Keywords: Dairy animal milks; Fat globules; Fatty acid; Human milk; Proteomics.
© 2025 The Authors. Published by Elsevier Ltd.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures


References
-
- Chardigny J.-M., Destaillats F., Malpuech-Brugère C., Moulin J., Bauman D.E., Lock A.L.…Chaumont P. Do trans fatty acids from industrially produced sources and from natural sources have the same effect on cardiovascular disease risk factors in healthy subjects? Results of the trans fatty acids collaboration (TRANSFACT) study. The American Journal of Clinical Nutrition. 2008;87(3):558–566. doi: 10.1093/ajcn/87.3.558. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

