Sexual Size Dimorphism Correlates With the Number of Androgen Response Elements in Mammals, But Only in Small-Bodied Species
- PMID: 40248910
- PMCID: PMC12015095
- DOI: 10.1093/gbe/evaf068
Sexual Size Dimorphism Correlates With the Number of Androgen Response Elements in Mammals, But Only in Small-Bodied Species
Abstract
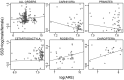
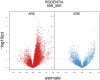
Sexual size dimorphism is common throughout the animal kingdom, but its evolution and development remain difficult to explain given most of the genome is shared between males and females. Sex-biased regulation of genes via sex hormone signaling offers an intuitive mechanism by which males and females could develop different body sizes. One prediction of this hypothesis is that the magnitude of sexual size dimorphism scales with the number of androgen response elements or estrogen response elements, the DNA motifs to which sex hormone receptors bind. Here, we test this hypothesis using 268 mammalian species with full genome assemblies and annotations. We find that in the two smallest-bodied lineages (Chiroptera and Rodentia), sexual size dimorphism increases (male-larger) as the number of androgen response elements in a genome increases. In fact, myomorph rodents-which are especially small-bodied with high sexual size dimorphism-show an explosion of androgen receptor elements in their genomes. In contrast, the three large-bodied lineages (orders Carnivora, Cetartiodactyla, and Primates) do not show this relationship, instead following Rensch's Rule, or the observation that sexual size dimorphism increases with overall body size. One hypothesis to unify these observations is that small-bodied organisms like bats and rodents tend to reach peak reproductive fitness quickly and are more reliant on hormonal signaling to achieve sexual size dimorphism over relatively short time periods. Our study uncovers a previously unappreciated relationship between sexual size dimorphism, body size, and hormone signaling that likely varies in ways related to life history.
Keywords: androgen signaling; evolution; genetics; hormone response elements; sexual size dimorphism.
© The Author(s) 2025. Published by Oxford University Press on behalf of Society for Molecular Biology and Evolution.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
A test of Rensch's rule in greater horseshoe bat (Rhinolophus ferrumequinum) with female-biased sexual size dimorphism.PLoS One. 2014 Jan 20;9(1):e86085. doi: 10.1371/journal.pone.0086085. eCollection 2014. PLoS One. 2014. PMID: 24465886 Free PMC article.
-
Sexual size dimorphism in lizards: Rensch's rule, reproductive mode, clutch size, and line fitting method effects.Integr Zool. 2022 Sep;17(5):787-803. doi: 10.1111/1749-4877.12569. Epub 2021 Jul 28. Integr Zool. 2022. PMID: 34216109
-
The relationship between sexual dimorphism and androgen response element proliferation in primate genomes.Evolution. 2022 Jun;76(6):1331-1346. doi: 10.1111/evo.14483. Epub 2022 Apr 25. Evolution. 2022. Retraction in: Evolution. 2024 Jan 4;78(1):209. doi: 10.1093/evolut/qpad182. PMID: 35420699 Free PMC article. Retracted.
-
Sexual dimorphism through androgen signaling; from external genitalia to muscles.Front Endocrinol (Lausanne). 2022 Jul 27;13:940229. doi: 10.3389/fendo.2022.940229. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35983512 Free PMC article. Review.
-
Gene Duplication of Androgen Receptor As An Evolutionary Driving Force Underlying the Diversity of Sexual Characteristics in Teleost Fishes.Zoolog Sci. 2024 Feb;41(1):68-76. doi: 10.2108/zs230098. Zoolog Sci. 2024. PMID: 38587519 Review.
References
-
- Badyaev A. Growing apart: an ontogenetic perspective on the evolution of sexual size dimorphism. Trends Ecol Evol. 2002:17:369–378. 10.1016/s0169-5347(02)02569-7. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

