Antitumor CD4+ T Helper 1 Cells Target and Control the Outgrowth of Disseminated Cancer Cells
- PMID: 40249209
- PMCID: PMC12046335
- DOI: 10.1158/2326-6066.CIR-24-0630
Antitumor CD4+ T Helper 1 Cells Target and Control the Outgrowth of Disseminated Cancer Cells
Abstract
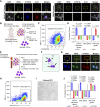
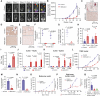
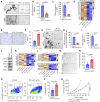
Detection of disseminated cancer cells (DCC) in the bone marrow (BM) of patients with breast cancer is a critical predictor of late recurrence and distant metastasis. Conventional therapies often fail to completely eradicate DCCs in patients. In this study, we demonstrate that intratumoral priming of antitumor CD4+ T helper 1 (Th1) cells was able to eliminate the DCC burden in distant organs and prevent overt metastasis, independent of CD8+ T cells. Intratumoral priming of tumor antigen-specific CD4+ Th1 cells enhanced their migration to the BM and distant metastatic site to selectively target DCC burden. The majority of these intratumorally activated CD4+ T cells were CD4+PD1- T cells, supporting their nonexhaustion stage. Phenotypic characterization revealed enhanced infiltration of memory CD4+ T cells and effector CD4+ T cells in the primary tumor, tumor-draining lymph node, and DCC-driven metastasis site. A robust migration of CD4+CCR7+CXCR3+ Th1 cells and CD4+CCR7-CXCR3+ Th1 cells into distant organs further revealed their potential role in eradicating DCC-driven metastasis. The intratumoral priming of antitumor CD4+ Th1 cells failed to eradicate DCC-driven metastasis in CD4- or IFN-γ knockout mice. Moreover, antitumor CD4+ Th1 cells, by increasing IFN-γ production, inhibited various molecular aspects and increased classical and nonclassical MHC molecule expression in DCCs. This reduced stemness and self-renewal while increasing immune recognition in DCCs of patients with breast cancer. These results unveil an immune basis for antitumor CD4+ Th1 cells that modulate DCC tumorigenesis to prevent recurrence and metastasis in patients.
©2025 The Authors; Published by the American Association for Cancer Research.
Conflict of interest statement
G. Ramamoorthi reports a patent for PCT/US2024/013114 pending to Moffitt Cancer Center. M.C. Lee reports other support from private donors during the conduct of the study. B.J. Czerniecki reports grants from CDMRP/DOD, Pennies in Action, and CDMRP/DOD during the conduct of the study as well as other support from ImmunoRestoration outside the submitted work and has a patent for use of syringe ready type I dendritic cells pending. No disclosures were reported by the other authors.
Figures




Similar articles
-
Sustained antigen-specific antitumor recall response mediated by gene-modified CD4+ T helper-1 and CD8+ T cells.Cancer Res. 2007 Dec 1;67(23):11428-37. doi: 10.1158/0008-5472.CAN-07-1141. Cancer Res. 2007. PMID: 18056471
-
Anti-HER2 CD4(+) T-helper type 1 response is a novel immune correlate to pathologic response following neoadjuvant therapy in HER2-positive breast cancer.Breast Cancer Res. 2015 May 23;17(1):71. doi: 10.1186/s13058-015-0584-1. Breast Cancer Res. 2015. PMID: 25997452 Free PMC article.
-
Myeloid-derived suppressor cells attenuate TH1 development through IL-6 production to promote tumor progression.Cancer Immunol Res. 2013 Jul;1(1):64-76. doi: 10.1158/2326-6066.CIR-13-0030. Epub 2013 Apr 29. Cancer Immunol Res. 2013. PMID: 24777249
-
Disseminated cancer cells in breast cancer: Mechanism of dissemination and dormancy and emerging insights on therapeutic opportunities.Semin Cancer Biol. 2022 Jan;78:78-89. doi: 10.1016/j.semcancer.2021.02.004. Epub 2021 Feb 21. Semin Cancer Biol. 2022. PMID: 33626407 Review.
-
Tumor antigen-specific T helper cells in cancer immunity and immunotherapy.Cancer Immunol Immunother. 2005 Aug;54(8):721-8. doi: 10.1007/s00262-004-0653-2. Epub 2005 Jan 27. Cancer Immunol Immunother. 2005. PMID: 16010587 Free PMC article. Review.
Cited by
-
Intratumoral Immunotherapy in Breast Cancer.Vaccines (Basel). 2025 Apr 19;13(4):429. doi: 10.3390/vaccines13040429. Vaccines (Basel). 2025. PMID: 40333343 Free PMC article. Review.
References
-
- Braun S, Vogl FD, Naume B, Janni W, Osborne MP, Coombes RC, et al. . A pooled analysis of bone marrow micrometastasis in breast cancer. N Engl J Med 2005;353:793–802. - PubMed
-
- Hartkopf AD, Brucker SY, Taran FA, Harbeck N, von Au A, Naume B, et al. . Disseminated tumour cells from the bone marrow of early breast cancer patients: results from an international pooled analysis. Eur J Cancer 2021;154:128–37. - PubMed
-
- Hartkopf AD, Wallwiener M, Fehm TN, Hahn M, Walter CB, Gruber I, et al. . Disseminated tumor cells from the bone marrow of patients with nonmetastatic primary breast cancer are predictive of locoregional relapse. Ann Oncol 2015;26:1155–60. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

