Small-scale fed-batch cultivations of Vibrio natriegens: overcoming challenges for early process development
- PMID: 40249449
- PMCID: PMC12089209
- DOI: 10.1007/s00449-025-03159-9
Small-scale fed-batch cultivations of Vibrio natriegens: overcoming challenges for early process development
Abstract
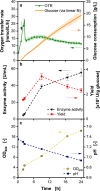
Vibrio natriegens is a fast-growing microbial workhorse with high potential for biotechnological applications. However, handling the bacterium in batch processes is challenging due to its high overflow metabolism and mixed acid formation under microaerobic conditions. For early process development, technologies enabling small-scale fed-batch cultivation of V. natriegens Vmax are needed. In this study, fed-batch cultivations in 96-well microtiter plates were successfully online-monitored for the first time with a µTOM device. Using the online-monitored oxygen transfer rate, a scale up to membrane-based fed-batch shake flasks was performed. The overflow metabolism was efficiently minimized by choosing suitable feed rates, and mixed acid formation was prevented. A glucose soft sensor using the oxygen transfer rate provided accurate estimates of glucose consumption throughout the fermentation, eliminating the need for offline sampling. Analyzing the impact of the inducer IPTG on the recombinant production of the enzyme inulosucrase revealed concentration-dependent effects in batch processes. In contrast, fed-batch operating mode resulted in high inulosucrase activity even without induction. Overall, an inulosucrase titer of 80 U/mL was achieved. In conclusion, the advantages of small-scale fed-batch technologies supported by a glucose soft sensor have been demonstrated for early process development for V. natriegens Vmax.
Keywords: Vibrio natriegens; Fed-batch; Glucose soft sensor; Microtiter plate.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: The authors declare no competing interests.
Figures

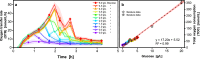
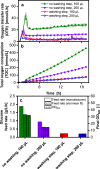
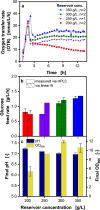
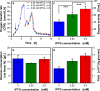

References
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources

