Suppressed intestinal secondary bile acids in moxifloxacin-induced hyperglycemia: studies in normal and diabetic GK rats
- PMID: 40255568
- PMCID: PMC12006139
- DOI: 10.3389/fphar.2025.1569856
Suppressed intestinal secondary bile acids in moxifloxacin-induced hyperglycemia: studies in normal and diabetic GK rats
Abstract
Objective: Moxifloxacin (MFLX) frequently induces dysglycemia when used in the treatment of infectious diseases, particularly in patients with diabetes. However, the mechanism through which MFLX affects host glucose metabolism remains unclear. This study aimed to investigate the possible mechanism underlying MFLX-induced hyperglycemia.
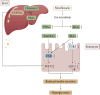
Methods: In this study, we investigated the short-term (3 days) and long-term (14 days) effects of MFLX on glucose metabolism in normal and type 2 diabetic GK rats. After oral administration of 40 mg/kg of MFLX, blood glucose, insulin, GLP-1, and fibroblast growth factor 15 (FGF15) levels in the blood of rats, as well as bile acids in both blood and feces, and gut microbiota, were examined. Liver and ileum tissues were promptly harvested for detecting the expression of hepatic 7α-hydroxylase (CYP7A1) and intestinal Takeda G-protein-coupled receptor 5 (TGR5) and farnesoid X receptor (FXR). In addition, we explored the effect of secondary bile acids (SBAs) on GLP-1 secretion in NCI-H716 cells, and observed the direct effect of MFLX on the expression of CYP7A1 in HepG2 cells and TGR5, FXR in NCI-H716 cells.
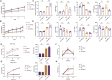
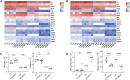
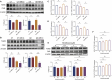
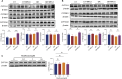
Results: It was demonstrated that MFLX induced hyperglycemia in diabetic rats, with a more pronounced reduction in serum insulin, GLP-1, and FGF15 levels than observed in normal rats. Gut microbiota associated with SBAs metabolism were significantly reduced, leading to decreased intestinal deoxycholic acid (DCA) and lithocholic acid (LCA). In vitro studies revealed that DCA and LCA (25 μM, 50 μM, and 100 μM) promoted GLP-1 secretion in a concentration-dependent manner in NCI-H716 cells. Meanwhile, we observed that the expression of intestinal TGR5 and FXR significantly downregulated, whereas CYP7A1 expression in liver was increased in GK rats after MFLX treatment. MFLX itself (0.1 μM, 1 μM, and 10 μM) did not directly altered TGR5 or FXR expressions in NCI-H716 cells, nor did it alter CYP7A1 expression in HepG2 cells, which indicated that the impact of MFLX on glucose metabolism was primarily induced by changes in bile acids metabolism resulting from alterations in the gut microbiota.
Conclusion: Our studies showed MFLX more likely to cause hyperglycemia when used in diabetic states and highlighted the critical role of gut microbiota-SBAs-TGR5/FXR pathway in MFLX-induced hyperglycemia.
Keywords: GLP-1; bile acid; gut microbiota; hyperglycemia; moxifloxacin.
Copyright © 2025 Sun, Qu, Yang, Lv, Wang, Fan, Wang, Pan, Du and Yu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Intestine farnesoid X receptor agonist and the gut microbiota activate G-protein bile acid receptor-1 signaling to improve metabolism.Hepatology. 2018 Oct;68(4):1574-1588. doi: 10.1002/hep.29857. Epub 2018 May 21. Hepatology. 2018. PMID: 29486523 Free PMC article.
-
Bile acids contribute to the development of non-alcoholic steatohepatitis in mice.JHEP Rep. 2021 Oct 13;4(1):100387. doi: 10.1016/j.jhepr.2021.100387. eCollection 2022 Jan. JHEP Rep. 2021. PMID: 34825156 Free PMC article.
-
Jiang-Tang-San-Huang pill alleviates type 2 diabetes mellitus through modulating the gut microbiota and bile acids metabolism.Phytomedicine. 2023 May;113:154733. doi: 10.1016/j.phymed.2023.154733. Epub 2023 Feb 26. Phytomedicine. 2023. PMID: 36870307
-
Bile acid nuclear receptor FXR and digestive system diseases.Acta Pharm Sin B. 2015 Mar;5(2):135-44. doi: 10.1016/j.apsb.2015.01.004. Epub 2015 Feb 25. Acta Pharm Sin B. 2015. PMID: 26579439 Free PMC article. Review.
-
Bile acids are nutrient signaling hormones.Steroids. 2014 Aug;86:62-8. doi: 10.1016/j.steroids.2014.04.016. Epub 2014 May 10. Steroids. 2014. PMID: 24819989 Free PMC article. Review.
References
-
- Bartoletti R., Claps F., Tulone G., Perotti A., Zucchi A., Riccardi N., et al. (2022). Antibiotic prophylaxis in patients who had undergone to prostate biopsy in between the EMA warning era: effects of fluoroquinolones in diabetic and non-diabetic patients. Results of an observational cohort study. World J. Urology 40, 2025–2031. 10.1007/s00345-022-04055-7 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

