Thalidomide augments maturation and T helper 1-inducing capacity of monocyte-derived dendritic cells in vitro
- PMID: 40256218
- PMCID: PMC12008494
- DOI: 10.34172/bi.30588
Thalidomide augments maturation and T helper 1-inducing capacity of monocyte-derived dendritic cells in vitro
Abstract
Introduction: Dendritic cells (DCs) possess specialized abilities to present antigens and stimulate T cells, making them essential in triggering adaptive immune responses. Thalidomide and its derivatives are classified as a group of medications that possess immunomodulatory properties. Numerous studies have demonstrated the contentious impact of these drugs on DCs. Therefore, the objective of the present study was to assess the influence of Thalidomide therapy on the maturation and stimulation of monocyte-derived DCs, and subsequently examine the consequences of these treated DCs on the immune responses of autologous T cells.
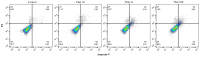
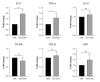
Methods: The immature DCs derived from monocytes were subjected to exposure to Thalidomide and Lipopolysaccharides (LPS) on the fifth day of differentiation, followed by a 24-hour incubation period. On the sixth day, the phenotypic features of the DCs in both the control and treatment groups were assessed using flow cytometry. Subsequently, the gene expression in both the DCs and autologous T cells co-cultured with the DCs was evaluated using the real-time PCR method.
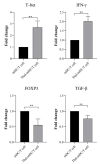
Results: Thalidomide-treated DCs exhibited a significant augmentation in the expression of maturation and stimulatory surface markers CD11c, HLA-DR, and CD86 (P ≤ 0.01), as well as gene expression of TNF-α and IL-12 (P ≤ 0.01) when compared to the control group. Furthermore, co-culture of Thalidomide-treated DCs with T cells increased T-bet and IFN-γ (P ≤ 0.01) expression, while diminished FOXP3 and TGF-β (P ≤ 0.01) expression compared to T cells co-cultured with untreated DCs.
Conclusion: Our findings indicate that in vitro Thalidomide treatment shifts DCs towards an immunogenic state and elevates their T helper 1 inducing capacity, which may be efficient in immunotherapy of various cancers.
Keywords: Cancer; Immunomodulation; Monocyte-derived dendritic cell; T cell; Thalidomide.
© 2025 The Author(s).
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Similar articles
-
Dopamine receptor agonist cabergoline promotes immunogenic phenotype in human monocyte-derived dendritic cells.Cell Biochem Funct. 2024 Jun;42(4):e4067. doi: 10.1002/cbf.4067. Cell Biochem Funct. 2024. PMID: 38874324
-
Phenotypic profile of dendritic and T cells in the lymph node of Balb/C mice with breast cancer submitted to dendritic cells immunotherapy.Immunol Lett. 2016 Sep;177:25-37. doi: 10.1016/j.imlet.2016.07.009. Epub 2016 Jul 14. Immunol Lett. 2016. PMID: 27423825
-
siRNA-Mediated B7H7 Knockdown in Gastric Cancer Lysate-Loaded Dendritic Cells Amplifies Expansion and Cytokine Secretion of Autologous T Cells.Biomedicines. 2023 Dec 4;11(12):3212. doi: 10.3390/biomedicines11123212. Biomedicines. 2023. PMID: 38137433 Free PMC article.
-
Everolimus treatment enhances inhibitory immune checkpoint molecules' expression in monocyte-derived dendritic cells.Hum Immunol. 2024 May;85(3):110798. doi: 10.1016/j.humimm.2024.110798. Epub 2024 Apr 2. Hum Immunol. 2024. PMID: 38569354
-
Characterization of canine monocyte-derived dendritic cells with phenotypic and functional differentiation.Can J Vet Res. 2007 Jul;71(3):165-74. Can J Vet Res. 2007. PMID: 17695590 Free PMC article.
References
-
- Masoumi J, Zainodini N, Basirjafar P, Tavakoli T, Zandvakili R, Nemati M, et al. Apelin receptor antagonist boosts dendritic cell vaccine efficacy in controlling angiogenic, metastatic and apoptotic-related factors in 4T1 breast tumor-bearing mice. Med Oncol. 2023;40:179. doi: 10.1007/s12032-023-02030-9. - DOI - PubMed
-
- Ghorbaninezhad F, Masoumi J, Bakhshivand M, Baghbanzadeh A, Mokhtarzadeh A, Kazemi T, et al. CTLA-4 silencing in dendritic cells loaded with colorectal cancer cell lysate improves autologous T cell responses in vitro. Front Immunol. 2022;13:931316. doi: 10.3389/fimmu.2022.931316. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials