Evaluating the safety, pharmacokinetics and efficacy of phage therapy in treating fracture-related infections with multidrug-resistant Staphylococcus aureus: intravenous versus local application in sheep
- PMID: 40256450
- PMCID: PMC12006137
- DOI: 10.3389/fcimb.2025.1547250
Evaluating the safety, pharmacokinetics and efficacy of phage therapy in treating fracture-related infections with multidrug-resistant Staphylococcus aureus: intravenous versus local application in sheep
Abstract
Background: Fracture-related infections (FRI), particularly those caused by antibiotic resistant Staphylococcus aureus, present significant clinical challenges due to the formation of biofilm on the implanted device, and reduced options for conventional antibiotic treatment. Bacteriophage (phage) therapy (PT) offers a targeted approach to managing such infections, however, evidence for pharmacokinetics and optimal route of administration is limited for FRI. This study aimed to evaluate safety, phage distribution kinetics, phage neutralization, and antibacterial efficacy after intravenous or local administration in a sheep model.
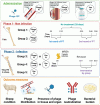
Methods: The study was conducted in two phases: Phase 1 assessed the safety and distribution of two successive rounds of intravenous and local phage administration in non-infected sheep, while Phase 2 evaluated the therapeutic efficacy of intravenous versus local phage administration in combination with intravenous vancomycin in treating MRSA-induced FRI (tibial osteotomy with plate fixation). The specific pathogen and phage used in the sheep were both taken from a human FRI patient treated with PT. Phage neutralization and phage distribution were the primary outcomes measured in both phases of the sheep study.
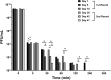
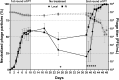
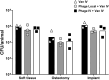
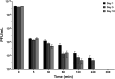
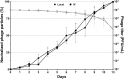
Results: Both intravenous and local phage administration were well-tolerated in non-infected sheep. Phages were cleared rapidly from circulation after intravenous administration, with no phage detected after 240 minutes. Phage neutralization increased during PT, peaking at 99.9% in non-inoculated sheep by the end of the second phage treatment (day 50). In infected sheep, phage neutralization levels reached a maximum of 99.9% earlier (day 13), with no significant differences between intravenous and local administration. The bacterial load was not significantly changed by PT, either IV or locally applied.
Conclusions: PT is a safe adjunct to antibiotic treatment for FRI, however, phage neutralization developed rapidly and was accelerated in infected hosts. Further research is required to optimize phage selection, dosing, and delivery methods to enhance its therapeutic potential as an adjunct to conventional antibiotic therapy, particularly in the face of challenges such as rapid clearance and phage neutralization.
Keywords: MRSA; Staphylococcus aureus; administration; bacteriophage; fracture related infection; neutralization; osteomyelitis; pharmacokinetics.
Copyright © 2025 Peez, Chen, Henssler, Chittò, Onsea, Verhofstad, Arens, Constant, Zeiter, Obremskey, Trampuz, Raschke, Zalavras, Metsemakers and Moriarty.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The handling editor MM declared a past co-authorship with the authors.
Figures
References
-
- Cano E. J., Caflisch K. M., Bollyky P. L., Van Belleghem J. D., Patel R., Fackler J., et al. (2021). Phage therapy for limb-threatening prosthetic knee Klebsiella pneumoniae infection: case report and in vitro characterization of anti-biofilm activity. Clin. Infect. Dis. 73, e144–ee51. doi: 10.1093/cid/ciaa705 - DOI - PMC - PubMed
-
- Chen B., Benavente L. P., Chitto M., Wychowaniec J. K., Post V., D’Este M., et al. (2023). Alginate microbeads and hydrogels delivering meropenem and bacteriophages to treat Pseudomonas aeruginosa fracture-related infections. J. Control Release 364, 159–173. doi: 10.1016/j.jconrel.2023.10.029 - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

