Discovery of Novel Multiangiogenic Agents Targeting VEGFR2, EphB4, FGFR-1, and TIE-2: Receptor-Based Pharmacophore Modeling, Virtual Screening, and Molecular Modeling Studies
- PMID: 40256504
- PMCID: PMC12004182
- DOI: 10.1021/acsomega.4c08366
Discovery of Novel Multiangiogenic Agents Targeting VEGFR2, EphB4, FGFR-1, and TIE-2: Receptor-Based Pharmacophore Modeling, Virtual Screening, and Molecular Modeling Studies
Abstract
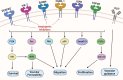
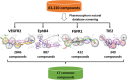
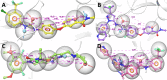
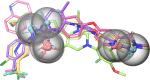
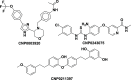
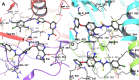
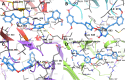
The angiogenesis phenomenon is crucial for the formation of new blood vessels in cancer cells. The cancerous cells' progress hampers other healthy cells. The main objective of this study is to explore and decipher multimodal natural compounds against VEGFR2, EphB4, FGFR-1, and TIE-2 drug targets to arrest angiogenesis and progression. The receptor-based pharmacophore modeling of VEGFR2, EphB4, FGFR-1, and TIE-2 was developed and validated through enrichment parameters. Further, the validated hypothesis allowed for screening druglike natural product databases such as SuperNatural 3.0, COCONUT, and LOTUS. The common pharmacophoric featured natural compounds were assessed for binding affinities using absolute end-point methods. Finally, density functional theory has been studied to understand the chemical reactivity and stability of the protein complexes. Among all of the screened natural compounds, 17 natural compounds were found to align accurately against validated pharmacophore models having higher fitness scores and align scores. Taking reference drugs sorafenib (VEGFR2), NVP-BHG712 (EphB4), pemiganitib (FGFR-1), and DP1919 (TIE-2), three promising natural compounds CNP0003920, CNP0243075, and CNP0211397 were concluded based on their end-point binding energies, binding interactions, molecular dynamics, and optimal pharmacokinetic and toxicity profiles. The density functional theory (DFT) results suggested that the identified compounds bound with protein complexes are stable. Our findings can represent a promising starting point for developing multimodal analogues VEGFR2, EphB4, FGFR-1, and TIE-2 proteins.
© 2025 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures







Similar articles
-
Molecular dynamics guided insight, binding free energy calculations and pharmacophore-based virtual screening for the identification of potential VEGFR2 inhibitors.J Recept Signal Transduct Res. 2019 Oct-Dec;39(5-6):415-433. doi: 10.1080/10799893.2019.1690509. Epub 2019 Nov 22. J Recept Signal Transduct Res. 2019. PMID: 31755336
-
Discovery of dual kinase inhibitors targeting VEGFR2 and FAK: structure-based pharmacophore modeling, virtual screening, and molecular docking studies.BMC Chem. 2024 Feb 12;18(1):29. doi: 10.1186/s13065-024-01130-5. BMC Chem. 2024. PMID: 38347617 Free PMC article.
-
Ligand-Based Pharmacophore Modeling, Molecular Docking, and Molecular Dynamic Studies of Dual Tyrosine Kinase Inhibitor of EGFR and VEGFR2.Int J Mol Sci. 2020 Oct 21;21(20):7779. doi: 10.3390/ijms21207779. Int J Mol Sci. 2020. PMID: 33096664 Free PMC article.
-
Exploring natural products as multi-target-directed drugs for Parkinson's disease: an in-silico approach integrating QSAR, pharmacophore modeling, and molecular dynamics simulations.J Biomol Struct Dyn. 2024;42(20):11167-11184. doi: 10.1080/07391102.2023.2260879. Epub 2023 Sep 27. J Biomol Struct Dyn. 2024. PMID: 37753798
-
Pharmacophore-Based Virtual Screening, Quantum Mechanics Calculations, and Molecular Dynamics Simulation Approaches Identified Potential Natural Antiviral Drug Candidates against MERS-CoV S1-NTD.Molecules. 2021 Aug 17;26(16):4961. doi: 10.3390/molecules26164961. Molecules. 2021. PMID: 34443556 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
