Intraepineurial Fat Fraction: A Novel MR Neurography-Based Biomarker in Transthyretin Amyloidosis Polyneuropathy
- PMID: 40256985
- PMCID: PMC12010196
- DOI: 10.1111/ene.70168
Intraepineurial Fat Fraction: A Novel MR Neurography-Based Biomarker in Transthyretin Amyloidosis Polyneuropathy
Abstract
Introduction: Hereditary transthyretin amyloid polyneuropathy (ATTRv-PN) is a rare and progressive neurodegenerative disorder characterized by axonal neuropathy and amyloid deposits. Early detection of disease onset and progression is crucial for timely therapeutic intervention. Quantitative MRI (qMRI) can be used to measure potential biomarkers. Intraepineurial fat fraction (ieFF) may reflect lipid droplets in amyloid deposits as described in histological studies or the replacement of nerve fiber loss with fatty-rich interfascicular epineurium. This study investigates the potential utility of ieFF as a novel imaging-related biomarker in differentiating ATTRv-PN, asymptomatic carriers (ATTRv-C), and healthy controls (HCs).
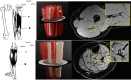
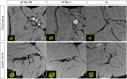
Methods: Fifty-three patients with TTR mutations were imaged (31 ATTRv-PN patients, 22 ATTRv-C, and 24 HC) and both clinical and electrophysiological parameters were quantified. 3D volume, ieFF, and magnetization transfer ratio (MTR) were quantified in sciatic and tibial nerves using qMRI.
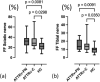
Results: Symptomatic ATTRv-PN patients exhibited significantly higher ieFF in both sciatic (32.4% IQR [24.4-38.1]) and tibial nerves (13.7%, IQR [9.97-20.7]) compared to controls (sciatic 22.3%, IQR [16.6-28.5]; tibial 9.74%, IQR [6.36-12.5]) (p < 0.05). ieFF values were positively correlated in both uni and multivariate analyses with the main clinical scores and electrophysiological measures. ATTRv-C also showed increased ieFF values compared to controls (p < 0.05). Comparatively, MTR and nerve volumes exhibited less pronounced differences across groups.
Conclusion: This study demonstrates that ieFF effectively differentiates symptomatic and asymptomatic ATTRv patients from HC and correlates strongly with electrophysiological and clinical severity parameters. Furthermore, we compare ieFF with conventional qMRI biomarkers, highlighting its superior potential for monitoring nerve structural impairment.
Keywords: biomarkers; fat fraction; neurography; quantified MRI; transthyretin amyloid polyneuropathy.
© 2025 The Author(s). European Journal of Neurology published by John Wiley & Sons Ltd on behalf of European Academy of Neurology.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Magnetization transfer ratio quantifies polyneuropathy in hereditary transthyretin amyloidosis.Ann Clin Transl Neurol. 2020 May;7(5):799-807. doi: 10.1002/acn3.51049. Epub 2020 Apr 25. Ann Clin Transl Neurol. 2020. PMID: 32333729 Free PMC article.
-
Magnetic resonance neurography and diffusion tensor imaging of the sciatic nerve in hereditary transthyretin amyloidosis polyneuropathy.J Neurol. 2023 Oct;270(10):4827-4840. doi: 10.1007/s00415-023-11813-z. Epub 2023 Jun 17. J Neurol. 2023. PMID: 37329346 Free PMC article.
-
In vivo detection of nerve injury in familial amyloid polyneuropathy by magnetic resonance neurography.Brain. 2015 Mar;138(Pt 3):549-62. doi: 10.1093/brain/awu344. Epub 2014 Dec 18. Brain. 2015. PMID: 25526974 Free PMC article.
-
The neuropathy in hereditary transthyretin amyloidosis: A narrative review.J Peripher Nerv Syst. 2021 Jun;26(2):155-159. doi: 10.1111/jns.12451. Epub 2021 May 11. J Peripher Nerv Syst. 2021. PMID: 33960565 Free PMC article. Review.
-
Hereditary transthyretin amyloidosis overview.Neurol Sci. 2022 Dec;43(Suppl 2):595-604. doi: 10.1007/s10072-020-04889-2. Epub 2020 Nov 14. Neurol Sci. 2022. PMID: 33188616 Free PMC article. Review.
References
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

