Enhancing glaucoma care with smart contact lenses: An overview of recent developments
- PMID: 40257617
- PMCID: PMC12011977
- DOI: 10.1007/s10544-025-00740-7
Enhancing glaucoma care with smart contact lenses: An overview of recent developments
Abstract
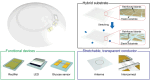
Glaucoma is a leading cause of irreversible blindness worldwide, affecting millions of individuals due to its progressive damage to the optic nerve, often caused by elevated intraocular pressure (IOP). Conventional methods of IOP monitoring, such as tonometry, provide sporadic and often inaccurate readings due to fluctuations throughout the day, leaving significant gaps in diagnosis and treatment. This review explores the transformative potential of smart contact lenses equipped with continuous IOP monitoring and therapeutic capabilities. These lenses integrate advanced materials such as graphene, nanogels, and magnetic oxide nanosheets alongside sophisticated biosensing and wireless communication systems. By offering continuous, real-time data, these lenses can detect subtle IOP fluctuations and provide immediate feedback to patients and clinicians. Moreover, drug-eluting capabilities embedded in these lenses present a groundbreaking approach to glaucoma therapy by improving medication adherence and providing controlled drug release directly to the eye. Beyond IOP management, these innovations also pave the way for monitoring biochemical markers and other ocular diseases. Challenges such as biocompatibility, long-term wearability, and affordability remain, but the integration of cutting-edge technologies in smart contact lenses signifies a paradigm shift in glaucoma care. These developments hold immense promise for advancing personalized medicine, improving patient outcomes, and mitigating the global burden of blindness.
Keywords: Biocompatible materials; Biosensors; Continuous monitoring; Drug delivery systems; Glaucoma; Intraocular pressure (IOP) monitoring; Ocular health; Smart contact lenses.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing Interests: The authors declare no competing interests.
Figures






Similar articles
-
Perioperative medications for preventing temporarily increased intraocular pressure after laser trabeculoplasty.Cochrane Database Syst Rev. 2017 Feb 23;2(2):CD010746. doi: 10.1002/14651858.CD010746.pub2. Cochrane Database Syst Rev. 2017. PMID: 28231380 Free PMC article.
-
Smart Contact Lens with High Sensitivity and Biocompatibility for Continuous Non-Invasive Intraocular Pressure Monitoring.ACS Sens. 2025 Jul 25;10(7):4996-5007. doi: 10.1021/acssensors.5c00883. Epub 2025 Jun 20. ACS Sens. 2025. PMID: 40540323
-
Effects and Safety of Press-Needle Therapy for Improving Visual Function and Eye Blood Circulation in Patients With Glaucoma With Controlled Intraocular Pressure: Study Protocol for a Multicenter Randomized Controlled Trial.JMIR Res Protoc. 2025 Apr 1;14:e67737. doi: 10.2196/67737. JMIR Res Protoc. 2025. PMID: 40168659 Free PMC article.
-
Genetic association of lipid traits and lipid-related drug targets with normal tension glaucoma: a Mendelian randomization study for predictive preventive and personalized medicine.EPMA J. 2024 Jul 13;15(3):511-524. doi: 10.1007/s13167-024-00373-5. eCollection 2024 Sep. EPMA J. 2024. PMID: 39239107
-
Cyclodestructive procedures for non-refractory glaucoma.Cochrane Database Syst Rev. 2018 Apr 25;4(4):CD009313. doi: 10.1002/14651858.CD009313.pub2. Cochrane Database Syst Rev. 2018. PMID: 29694684 Free PMC article.
References
-
- A.A. Abdulamier, L.M Shaker, A.A Al-Amiery, Advancements in the chemistry of contact lenses: Innovations and applications. Results in Chemistry, page 101872, (2024)
-
- M. Asplund, E. Thaning, J. Lundberg, A.C. Sandberg-Nordqvist, B. Kostyszyn, O. Inganäs, H. Von Holst, Toxicity evaluation of PEDOT/biomolecular composites intended for neural communication electrodes. Biomedical Materials. 4(4),045009, 7 (2009) - PubMed
-
- S. Asrani, R. Zeimer, J. Wilensky, D. Gieser, Susan Vitale, and Kim Lindenmuth. Large diurnal fluctuations in intraocular pressure are an independent risk factor in patients with glaucoma. Journal of Glaucoma. 9(2),134–142, 4 (2000) - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

