Physiological roles of embryonic microglia and their perturbation by maternal inflammation
- PMID: 40260079
- PMCID: PMC12009865
- DOI: 10.3389/fncel.2025.1552241
Physiological roles of embryonic microglia and their perturbation by maternal inflammation
Abstract
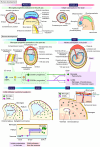
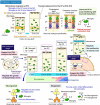
The interplay between the nervous and immune systems is well documented in the context of adult physiology and disease. Recent advances in understanding immune cell development have highlighted a significant interaction between neural lineage cells and microglia, the resident brain macrophages, during developmental stages. Throughout development, particularly from the embryonic to postnatal stages, diverse neural lineage cells are sequentially generated, undergo fate determination, migrate dynamically to their appropriate locations while maturing, and establish connections with their surroundings to form neural circuits. Previous studies have demonstrated that microglia contribute to this highly orchestrated process, ensuring the proper organization of brain structure. These findings underscore the need to further investigate how microglia behave and function within a broader framework of neurodevelopment. Importantly, recent epidemiological studies have suggested that maternal immune activation (MIA), triggered by various factors, such as viral or bacterial infections, environmental stressors, or other external influences, can affect neurogenesis and neural circuit formation, increasing the risk of neurodevelopmental disorders (NDDs) in offspring. Notably, many studies have revealed that fetal microglia undergo significant changes in response to MIA. Given their essential roles in neurogenesis and vascular development, inappropriate activation or disruption of microglial function may impair these critical processes, potentially leading to abnormal neurodevelopment. This review highlights recent advances in rodent models and human studies that have shed light on the behaviors and multifaceted roles of microglia during brain development, with a particular focus on the embryonic stage. Furthermore, drawing on insights from rodent MIA models, this review explores how MIA disrupts microglial function and how such disturbances may impair brain development, ultimately contributing to the onset of NDDs.
Keywords: brain; development; maternal immune activation; maternal inflammation; microglia; neurodevelopmental disorder; neuron; psychiatric disorder.
Copyright © 2025 Shimamura, Kitashiba, Nishizawa and Hattori.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources

