Factor XII-driven coagulation traps bacterial infections
- PMID: 40261297
- PMCID: PMC12013512
- DOI: 10.1084/jem.20250049
Factor XII-driven coagulation traps bacterial infections
Abstract
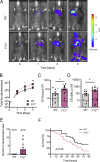
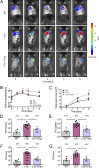
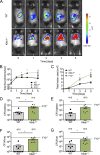
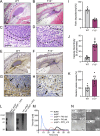
Blood coagulation is essential for stopping bleeding but also drives thromboembolic disorders. Factor XII (FXII)-triggered coagulation promotes thrombosis while being dispensable for hemostasis, making it a potential anticoagulant target. However, its physiological role remains unclear. Here, we demonstrate that FXII-driven coagulation enhances innate immunity by trapping pathogens and restricting bacterial infection in mice. Streptococcus pneumoniae infection was more severe in FXII-deficient (F12-/-) mice, with increased pulmonary bacterial burden, systemic spread, and mortality. Similarly, Staphylococcus aureus skin infections and systemic dissemination were exacerbated in F12-/- mice. Reconstitution with human FXII restored bacterial containment. Plasma kallikrein amplifies FXII activation, and its deficiency aggravated S. aureus skin infections, similarly to F12-/- mice. FXII deficiency impaired fibrin deposition in abscess walls, leading to leaky capsules and bacterial escape. Bacterial long-chain polyphosphate activated FXII, triggering fibrin formation. Deficiency in FXII substrate factor XI or FXII/factor XI co-deficiency similarly exacerbated S. aureus infection. The data reveal a protective role for FXII-driven coagulation in host defense, urging caution in developing therapeutic strategies targeting this pathway.
© 2025 Nickel et al.
Conflict of interest statement
Disclosures: K.F. Nickel reported being a current employee at Swedish Orphan Biovitrum GmbH. T. Renné reported patent application number: WO2006066878A1 (Renné, T. & Nieswandt, B., 2005, Targeting Factor XII for Prevention of Thrombus Formation and/or Stabilization.). No other disclosures were reported.
Figures


References
-
- Bochenska, O., Rapala-Kozik M., Wolak N., Kamysz W., Grzywacz D., Aoki W., Ueda M., and Kozik A.. 2015. Inactivation of human kininogen-derived antimicrobial peptides by secreted aspartic proteases produced by the pathogenic yeast Candida albicans. Biol. Chem. 396:1369–1375. 10.1515/hsz-2015-0167 - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

