Best Responders and Super-Responders to Calcipotriol and Betamethasone Dipropionate PAD-Cream: A Post Hoc Pooled Analysis of Two Phase 3 Trials
- PMID: 40274711
- PMCID: PMC12092894
- DOI: 10.1007/s13555-025-01418-x
Best Responders and Super-Responders to Calcipotriol and Betamethasone Dipropionate PAD-Cream: A Post Hoc Pooled Analysis of Two Phase 3 Trials
Abstract
Introduction: Individual endpoints from phase 3 trials demonstrated high efficacy and convenience for the calcipotriol/betamethasone dipropionate (CAL/BDP) cream based on polyaphron dispersion (PAD) technology compared to CAL/BDP gel. The objectives are to assess the proportion of best responders to CAL/BDP PAD-cream at weeks 4 and 8 and to identify the super-responders through the patients' key baseline characteristics associated with best response.
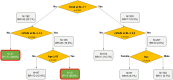
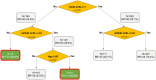
Methods: This was a post hoc pooled analysis of adult patients with mild-to-moderate psoriasis from two phase 3 trials: MC2-01-C2 (NCT03308799) and MC2-01-C7 (NCT03802344). Patients were randomly assigned (3:1:3) to CAL/BDP PAD-cream, PAD-cream vehicle, or CAL/BDP gel once daily. Best responders at weeks 4 and 8 were defined as patients achieving a restrictive endpoint comprising the combination of Physician's Global Assessment (PGA) controlled disease, modified Psoriasis Area and Severity Index (mPASI) success, and Dermatology Life Quality Index (DLQI) satisfaction. Best response rates were compared between treatments by logistic regression using multiple imputation. A classification and regression tree (CART) based on observed cases identified super-responders through the key baseline characteristics associated with best response among CAL/BDP PAD-cream users. All analyses were based on the modified intention-to-treat (mITT) population.
Results: The mITT included 551 patients on CAL/BDP PAD-cream, 542 on CAL/BDP gel, and 178 on vehicle. The CAL/BDP PAD-cream group showed a statistically significant higher best response rate compared to CAL/BDP gel at week 4 (10.7% vs 6.1%; p = 0.0048) and week 8 (27.4% vs 16.1%; p < 0.0001), and also compared to vehicle. Among CAL/BDP PAD-cream users, 63.6% of patients with a baseline DLQI < 7 and mPASI < 4.0 achieved best response at week 8 and were considered super-responders.
Conclusion: CAL/BDP PAD-cream demonstrated higher best response rates compared to CAL/BDP gel and vehicle in adults with mild-to-moderate psoriasis. Baseline DLQI and mPASI scores may predict which patients are most likely to achieve best response to CAL/BDP PAD-cream.
Trials registration: ClinicalTrials.gov identifiers, NCT03308799 (MC2-01-C2) and NCT03802344 (MC2-01-C7).
Keywords: Best responders; Betamethasone dipropionate; Calcipotriol; Psoriasis; Psoriasis treatment.
Plain language summary
This study evaluated a new calcipotriol and betamethasone dipropionate (CAL/BDP) cream with polyaphron dispersion (PAD) technology, designed to provide efficacy and safety with greater comfort for patients with mild-to-moderate psoriasis. The study compared the new cream to CAL/BDP gel and a dermatological vehicle to identify patients who responded best and which patient characteristics predicted best outcomes. Data from 1271 adult patients with mild-to-moderate psoriasis who participated in two trials were analyzed. Patients applied CAL/BDP PAD-cream, CAL/BDP gel, or vehicle once daily. Researchers defined “best responders” as patients achieving three key goals after 8 weeks of treatment: (1) clear or almost clear skin, (2) very low psoriasis severity, and (3) no significant impact on quality of life. They also identified “super-responders” as those most likely to achieve best outcomes, based on patient characteristics at the start of the study. CAL/BDP PAD-cream resulted in significantly more best responders compared to gel or vehicle. By week 4, 10.7% of cream users achieved best response compared to 6.1% of gel users. By week 8, these numbers rose to 27.4% for the cream and 16.1% for the gel. Among cream users, patients without an important affection of their quality of life and less severity at the start of the study had the highest likelihood of being super-responders. Overall, CAL/BDP PAD-cream was more effective than gel or vehicle in treating mild-to-moderate psoriasis. Patients with lower baseline disease severity were more likely to benefit the most.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of Interest: Andreas Pinter has no conflicts of interest to declare. Jordi Galván is an employee of Almirall S.A. Frank Freischläger is co-founder and co-owner of Estimondo GmbH. Ethical Approval: The study protocols of the two phase 3 studies were reviewed and approved by local institutional review boards or ethics committees (central institutional review board for trial MC2-01-C2: Quorum, 1501 Fourth Ave, Ste 800, Seattle, WA 98101; independent ethics committee for trial MC2-01-C7: Ethikkommission der Ärztekammer, Hamburg, Weidestraße 122b, 22083 Hamburg, Germany). The study was performed in accordance with the Helsinki Declaration of 1964 and its later amendments. All participants provided written informed to participate in the phase 3 studies.
Figures
Similar articles
-
Randomized Phase 3 trial demonstrating high efficacy, favourable safety and convenience of a novel calcipotriol and betamethasone dipropionate cream for the treatment of psoriasis.J Eur Acad Dermatol Venereol. 2023 Nov;37(11):2327-2335. doi: 10.1111/jdv.19330. Epub 2023 Aug 25. J Eur Acad Dermatol Venereol. 2023. PMID: 37432045 Clinical Trial.
-
Calcipotriene and Betamethasone Dipropionate PAD-Cream Demonstrates Greater Treatment Efficacy in Patients with Moderate-to-Severe Psoriasis Compared to Topical Suspension/Gel: A Subgroup Analysis of Two Phase 3 Studies.Dermatol Ther (Heidelb). 2023 Sep;13(9):2031-2044. doi: 10.1007/s13555-023-00979-z. Epub 2023 Jul 25. Dermatol Ther (Heidelb). 2023. PMID: 37490268 Free PMC article.
-
A novel, fixed-dose calcipotriol and betamethasone dipropionate cream for the topical treatment of plaque psoriasis: Direct and indirect evidence from phase 3 trials discussed at the 30th EADV Congress 2021.J Eur Acad Dermatol Venereol. 2023 Jan;37 Suppl 1:14-19. doi: 10.1111/jdv.18755. J Eur Acad Dermatol Venereol. 2023. PMID: 36546465 Review.
-
Pooled Analysis Demonstrating Superior Patient- Reported Psoriasis Treatment Outcomes for Calcipotriene/ Betamethasone Dipropionate Cream Versus Suspension/Gel.J Drugs Dermatol. 2022 Mar 1;21(3):242-248. doi: 10.36849/JDD.661. J Drugs Dermatol. 2022. PMID: 35254765 Clinical Trial.
-
Calcipotriol and Betamethasone Dipropionate Cream Based on PAD Technology for the Treatment of Plaque Psoriasis: A Narrative Review.Dermatol Ther (Heidelb). 2023 Oct;13(10):2153-2169. doi: 10.1007/s13555-023-01003-0. Epub 2023 Sep 23. Dermatol Ther (Heidelb). 2023. PMID: 37740858 Free PMC article. Review.
References
-
- AlQassimi S, AlBrashdi S, Galadari H, Hashim MJ. Global burden of psoriasis-comparison of regional and global epidemiology, 1990 to 2017. Int J Dermatol. 2020;59(5):566–71. - PubMed
-
- Bożek A, Reich A. The reliability of three psoriasis assessment tools: psoriasis area and severity index, body surface area and physician global assessment. Adv Clin Exp Med. 2017;26(5):851–6. - PubMed
-
- Manalo IF, Gilbert KE, Wu JJ. An updated survey for the 2007–2013 period of randomized controlled trials for psoriasis: treatment modalities, study designs, comparators, outcome measures and sponsorship. J Eur Acad Dermatol Venereol. 2015;29(10):1945–50. - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

