Preclinical validation of TGFβ inhibitors as a novel therapeutic strategy for post-traumatic heterotopic ossification
- PMID: 40274953
- PMCID: PMC12022333
- DOI: 10.1038/s41598-025-96961-0
Preclinical validation of TGFβ inhibitors as a novel therapeutic strategy for post-traumatic heterotopic ossification
Abstract
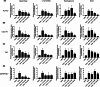
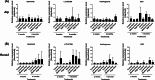
Heterotopic ossification (HO) is characterized by the abnormal growth of ectopic bone in non-skeletal soft tissues through a fibrotic pathway and is a frequent complication in a wide variety of musculoskeletal injuries. We have previously demonstrated that TGFβ levels are elevated in the soft tissues following extremity injuries. Since TGFβ mediates the initial inflammatory and wound-healing response in the traumatized muscle bed, we hypothesized that targeted inhibition of the TGFβ pathway may be able to abrogate the unbalanced fibrotic phenotype and bone-forming response observed in post-traumatic HO. Primary mesenchymal progenitor cells (MPCs) harvested from debrided traumatized human muscle tissue were used in this study. After treatment with TGFβ inhibitors (SB431542, Galunisertib/LY2157299, Halofuginone, and SIS3) cell proliferation/survival, fibrotic formation, osteogenic induction, gene expression, and phosphorylation of SMAD2/3 were assessed. In vivo studies were performed with a Sprague-Dawley rat blast model treated with the TGFβ inhibitors. The treatment effects on the rat tissues were investigated by radiographs, histology, and gene expression analyses. Primary MPCs treated with TGFβ had a significant increase in the number of fibrotic nodules compared to the control, while TGFβ inhibitors that directly block the TGFβ extracellular receptor had the greatest effect on reducing the number of fibrotic nodules and significantly reducing the expression of fibrotic genes. In vivo studies demonstrated a trend towards a lower extent of HO formation by radiographic analysis up to 4 months after injury when animals were treated with the TGFβ inhibitors SB431542, Halofuginone and SIS3. Altogether, our results suggest that targeted inhibition of the TGFβ pathway may be a useful therapeutic strategy for post-traumatic HO patients.
Keywords: Fibrosis; Heterotopic ossification; SMAD2; SMAD3; TGFβ; TGFβ inhibitors; Trauma.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Ethics approval and consent to participate: Primary MPCs were harvested from debrided traumatized human muscle tissue from injured service members during their initial surgical debridement at Walter Reed National Military Medical Center. Tissue specimens used in this study were taken at the margin of devitalized and healthy-appearing tissue that would otherwise be discarded as surgical waste. The Walter Reed National Military Medical Center Institutional Review Board approved this tissue procurement protocol and the need to obtain informed consent was waived by the Walter Reed National Military Medical Center Institutional Review Board. All experimental protocols were performed in accordance with relevant guidelines and regulations. Institutional animal care and use committee approval: All animal procedures were approved by the Institutional Animal Care and Use Committee at the Medical University of South Carolina. Animal experiments were performed in compliance with the “Animal Research: Reporting of In Vivo Experiments” (ARRIVE) guidelines ( https://arriveguidelines.org/ ). All animal experiments were performed in accordance with relevant guidelines and regulations.
Figures








References
-
- Potter, B. K. et al. Heterotopic ossification following traumatic and combat-related amputations. Prevalence, risk factors, and preliminary results of excision. J. Bone Joint Surg. Am.89 (3), 476–486 (2007). - PubMed
-
- Uezumi, A. et al. Fibrosis and adipogenesis originate from a common mesenchymal progenitor in skeletal muscle. J. Cell. Sci.124 (Pt 21), 3654–3664 (2011). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

