Single-nucleus RNA sequencing defines adipose tissue subpopulations that contribute to Tibetan pig cold adaptation
- PMID: 40275312
- PMCID: PMC12023645
- DOI: 10.1186/s12915-025-02211-0
Single-nucleus RNA sequencing defines adipose tissue subpopulations that contribute to Tibetan pig cold adaptation
Abstract
Background: Thermogenic beige adipocyte displays a remarkable ability in mammals to adapt to cold environments, but the underlying cellular mechanisms remain unclear, especially in pigs that lack functional UCP1.
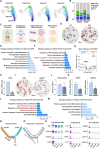
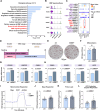
Results: Multilocular beige adipocytes were observed in both Tibetan pigs (cold-tolerant) and Bama pigs (cold-sensitive) after short-term cold exposure (4 ℃ for 3 days). Through single-nucleus RNA sequencing of adipose tissues, including subcutaneous inguinal adipose tissues (IAT) and perirenal adipose tissues (PAT), from both pig breeds at room temperature and cold treatment conditions, we discovered two cell subpopulations specific to Tibetan pigs, PDGFRα+EBF2High in IAT and ADIPOQ+HIF1AHigh in both depots. PDGFRα+EBF2High cells were characterized as potential beige precursors, while ADIPOQ+HIF1AHigh cells were found to express highly thermogenic-related genes. Despite the decrease of the lipogenic subpopulation and the increase of the lipolytic and the thermogenic subpopulations observed in both pig breeds upon cold treatment, Tibetan pigs exhibited stronger cellular and molecular responses compared to Bama pigs. Remarkably, cold-induced de novo beige adipogenesis and white adipocyte browning, likely occurred in Tibetan pigs, while Bama pigs relied more heavily on white browning. Moreover, BMP7, which was highly expressed in the PDGFRα+EBF2High subpopulation, positively regulates porcine beige thermogenic capacity.
Conclusions: Our data offers a comprehensive and unprecedented perspective on the heterogeneity and plasticity of adipose tissues of pigs and broadens the understanding of beige fat biology in mammals.
Keywords: Adipocytes; Cold stimulation; Pig; Subpopulations; snRNA-seq.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: All experiments involving pigs were conducted according to the guidelines for the care and use of laboratory animals established by the Beijing Association for Laboratory Animal Science and approved by the Animal Ethics Committee of the Institute of Animal Science (IAS2020 - 21), Chinese Academy of Agricultural Science. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures





Similar articles
-
Cold adaptation in pigs depends on UCP3 in beige adipocytes.J Mol Cell Biol. 2017 Oct 1;9(5):364-375. doi: 10.1093/jmcb/mjx018. J Mol Cell Biol. 2017. PMID: 28486585
-
Perirenal adipose tissue contains a subpopulation of cold-inducible adipocytes derived from brown-to-white conversion.Elife. 2024 Mar 12;13:RP93151. doi: 10.7554/eLife.93151. Elife. 2024. PMID: 38470102 Free PMC article.
-
Adaptive Thermogenesis and Lipid Metabolism Modulation in Inguinal and Perirenal Adipose Tissues of Hezuo Pigs in Response to Low-Temperature Exposure.Cells. 2025 Mar 7;14(6):392. doi: 10.3390/cells14060392. Cells. 2025. PMID: 40136641 Free PMC article.
-
Unraveling the complexity of thermogenic remodeling of white fat reveals potential antiobesity therapies.Genes Dev. 2021 Nov 1;35(21-22):1395-1397. doi: 10.1101/gad.349053.121. Genes Dev. 2021. PMID: 34725126 Free PMC article. Review.
-
Brown and Beige Adipose Tissues in Health and Disease.Compr Physiol. 2017 Sep 12;7(4):1281-1306. doi: 10.1002/cphy.c170001. Compr Physiol. 2017. PMID: 28915325 Free PMC article. Review.
References
-
- Cannon B, Nedergaard J. Brown adipose tissue: function and physiological significance. Physiol Rev. 2004;84(1):277–359. - PubMed
-
- Bartelt A, Heeren J. Adipose tissue browning and metabolic health. Nat Rev Endocrinol. 2014;10(1):24–36. - PubMed
-
- Chouchani ET, Kazak L, Spiegelman BM. New advances in adaptive thermogenesis: UCP1 and Beyond. Cell Metab. 2019;29(1):27–37. - PubMed
MeSH terms
Grants and funding
- No. 32025034 and No. 31925036/the National Natural Science Foundation for Distinguished Young Scholars
- No. 32025034 and No. 31925036/the National Natural Science Foundation for Distinguished Young Scholars
- U22A20507/the Joint Funds of the National Natural Science Foundation of China
- U22A20507/the Joint Funds of the National Natural Science Foundation of China
- No. 32230100 and No. 32330099/Key Program of National Natural Science of China
LinkOut - more resources
Full Text Sources
Miscellaneous

