Treatment with human placental extracts inhibits allergic rhinitis by modulating AMPK/SHP1/SHP2/STING signaling
- PMID: 40280106
- PMCID: PMC12059460
- DOI: 10.3892/mmr.2025.13548
Treatment with human placental extracts inhibits allergic rhinitis by modulating AMPK/SHP1/SHP2/STING signaling
Abstract
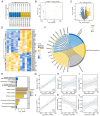
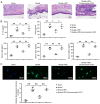
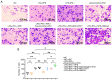
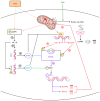
The present study aimed to investigate the regulatory effects and mechanisms of human placental extracts (HPE) on rats and cell models of ovalbumin (OVA)‑induced allergic rhinitis (AR). IFN‑y and LPS induced AR in vitro. A total of 32 male Sprague‑Dawley (SD) rats were randomly divided into the following four groups: Sham group, model group, model + HPE group and model + HPE + AMPK inhibitor group (n=8 rats/group). With the exception of the sham group, the remaining three groups were sensitized with OVA to establish an AR model, followed by various treatments. Hematoxylin and eosin staining was utilized to observe morphological changes in the nasal mucosa, ELISA was employed to measure serum levels of IL‑1β, interferon (IFN)β, immunoglobulin (Ig)E, IgG1 and IgG2a, and western blotting was conducted to assess protein expression across the groups. The sham group exhibited intact tissue structure with no notable pathological alterations. The model group demonstrated pronounced pathological features, including extensive infiltration of inflammatory cells, tissue shedding and edema. The model + HPE group revealed a gradual restoration of tissue architecture, characterized by reduced edema and inflammatory infiltration, whereas the model + HPE + AMPK inhibitor group again exhibited significant inflammatory cell infiltration and other pathological manifestations. Compared with the sham operation group, the levels of IL‑1β, IFNβ, IgE, IgG1 and IgG2a in the serum of the model group were elevated. The levels of IL‑1β, IFNβ, IgE, IgG1 and IgG2a in the model + HPE group were lower than those in the model group. In addition, the levels of IL‑1β, IFNβ, IgE, IgG1 and IgG2a in the model + HPE + AMPK inhibitor group were higher than those in the model + HPE group. Relative to the sham group, the expression levels of phosphorylated (p)‑AMPK/total (t)‑AMPK, p‑Src homology 2‑containing phosphatase (SHP)1/t‑SHP1 and p‑SHP2/t‑SHP2 were diminished, whereas the expression levels of p‑STING/t‑STING and p‑TBK1/t‑TBK1 were heightened in the model group. In comparison to the model group, the expression levels of p‑AMPK/t‑AMPK, p‑SHP1/t‑SHP1 and p‑SHP2/t‑SHP2 were enhanced, whereas the expression levels of p‑STING/t‑STING and p‑TBK1/t‑TBK1 were reduced in the model + HPE group. Conversely, when compared with the model + HPE group, the expression levels of p‑AMPK/t‑AMPK, p‑SHP1/t‑SHP1 and p‑SHP2/t‑SHP2 were decreased, whereas those of p‑STING/t‑STING and p‑TBK1/t‑TBK1 were increased in the model + HPE + AMPK inhibitor group. In conclusion, HPE may inhibit AR by modulating the AMPK/SHP1/SHP2/STING signaling pathway.
Keywords: AMPK/SHP1/SHP2/STING; allergic rhinitis; human placental extracts; ovalbumin.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
Acupuncture at "Die E acupoint" alleviates inflammatory reaction via inhibiting TLR4/MyD88/NF-κB signaling in rats with allergic rhinitis.Zhen Ci Yan Jiu. 2024 May 25;49(5):456-462. doi: 10.13702/j.1000-0607.20230724. Zhen Ci Yan Jiu. 2024. PMID: 38764116 Chinese, English.
-
Human placental extract reduces allergic inflammation in a murine allergic rhinitis model.Laryngoscope. 2014 Oct;124(10):E399-404. doi: 10.1002/lary.24714. Epub 2014 Jun 3. Laryngoscope. 2014. PMID: 24729028
-
Human placental extract regulates polarization of macrophages via IRGM/NLRP3 in allergic rhinitis.Biomed Pharmacother. 2023 Apr;160:114363. doi: 10.1016/j.biopha.2023.114363. Epub 2023 Feb 4. Biomed Pharmacother. 2023. PMID: 36746096
-
Saikosaponin A ameliorates nasal inflammation by suppressing IL-6/ROR-γt/STAT3/IL-17/NF-κB pathway in OVA-induced allergic rhinitis.Chem Biol Interact. 2020 Jan 5;315:108874. doi: 10.1016/j.cbi.2019.108874. Epub 2019 Oct 24. Chem Biol Interact. 2020. PMID: 31669322
-
Ozone inhalation induces exacerbation of eosinophilic airway inflammation and Th2-skew immune response in a rat model of AR.Biomed Pharmacother. 2021 May;137:111261. doi: 10.1016/j.biopha.2021.111261. Epub 2021 Jan 19. Biomed Pharmacother. 2021. PMID: 33482509
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

