Tamoxifen induces protection against manganese toxicity by REST upregulation via the ER-α/Wnt/β-catenin pathway in neuronal cells
- PMID: 40280417
- PMCID: PMC12152632
- DOI: 10.1016/j.jbc.2025.108529
Tamoxifen induces protection against manganese toxicity by REST upregulation via the ER-α/Wnt/β-catenin pathway in neuronal cells
Abstract
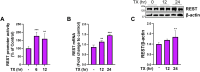
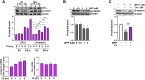
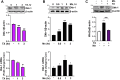
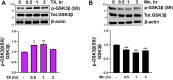
Chronic exposure to elevated levels of manganese (Mn) causes a neurological disorder referred to as manganism, with symptoms resembling Parkinson's disease (PD). The repressor element-1 silencing transcription factor (REST) has been shown to be neuroprotective in several neurological disorders, including PD, suggesting that identifying REST upregulation mechanisms is an important avenue for the development of novel therapeutics. 17β-estradiol (E2) activates the Wnt/β-catenin signaling, which is known to increase REST transcription. E2 and tamoxifen (TX), a selective estrogen receptor modulator, exerted protection against Mn toxicity. In this study, we tested if TX upregulates REST potentially via Wnt/β-catenin signaling in Cath.a-differentiated (CAD) neuronal cells using luciferase assay, qPCR, Western blot analysis, immunocytochemistry, mutagenesis, chromatin immunoprecipitation, and electrophoretic mobility shift assay. TX (1 μM) increased REST promoter activities and mRNA/protein levels and attenuated Mn (250 μM)-decreased REST transcription in parallel with TX's protective effects against Mn-induced toxicity, potentially via Wnt. TX activated Wnt/β-catenin signaling by preventing β-catenin degradation via inactivation of glycogen synthase kinase-3 beta, leading to increased β-catenin levels and its nuclear translocation and binding to T-cell factor/lymphoid enhancer binding factor sites on Wnt-responsive elements (WRE) of the REST promoter. Mutation of WRE abolished TX-induced REST promoter activity. TX-induced Wnt signaling activation was primarily via the estrogen receptor (ER)-α, although ER-β and G protein-coupled estrogen receptor 1 also mediated TX's action on REST transcription. These findings underscore the critical role of Wnt/β-catenin signaling in TX-induced REST transcription, affording protection mechanisms against Mn toxicity and neurological disorders associated with REST dysfunction.
Keywords: NRSF; REST; Wnt signaling; estrogen receptor; manganese; neuroprotection; tamoxifen; β-catenin.
Copyright © 2025 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interest The authors declare that they have no conflicts of interest with the contents of this article.
Figures







References
-
- Newland M.C., Ceckler T.L., Kordower J.H., Weiss B. Visualizing manganese in the primate basal ganglia with magnetic resonance imaging. Exp. Neurol. 1989;106:251–258. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

