Development and evaluation of a non-invasive brain-spine interface using transcutaneous spinal cord stimulation
- PMID: 40281628
- PMCID: PMC12023432
- DOI: 10.1186/s12984-025-01628-6
Development and evaluation of a non-invasive brain-spine interface using transcutaneous spinal cord stimulation
Abstract
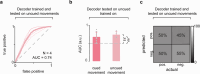
Motor rehabilitation is a therapeutic process to facilitate functional recovery in people with spinal cord injury (SCI). However, its efficacy is limited to areas with remaining sensorimotor function. Spinal cord stimulation (SCS) creates a temporary prosthetic effect that may allow further rehabilitation-induced recovery in individuals without remaining sensorimotor function, thereby extending the therapeutic reach of motor rehabilitation to individuals with more severe injuries. In this work, we report our first steps in developing a non-invasive brain-spine interface (BSI) based on electroencephalography (EEG) and transcutaneous spinal cord stimulation (tSCS). The objective of this study was to identify EEG-based neural correlates of lower limb movement in the sensorimotor cortex of unimpaired individuals (N = 17) and to quantify the performance of a linear discriminant analysis (LDA) decoder in detecting movement onset from these neural correlates. Our results show that initiation of knee extension was associated with event-related desynchronization in the central-medial cortical regions at frequency bands between 4 and 44 Hz. Our neural decoder using µ (8-12 Hz), low β (16-20 Hz), and high β (24-28 Hz) frequency bands achieved an average area under the curve (AUC) of 0.83 ± 0.06 s.d. (n = 7) during a cued movement task offline. Generalization to imagery and uncued movement tasks served as positive controls to verify robustness against movement artifacts and cue-related confounds, respectively. With the addition of real-time decoder-modulated tSCS, the neural decoder performed with an average AUC of 0.81 ± 0.05 s.d. (n = 9) on cued movement and 0.68 ± 0.12 s.d. (n = 9) on uncued movement. Our results suggest that the decrease in decoder performance in uncued movement may be due to differences in underlying cortical strategies between conditions. Furthermore, we explore alternative applications of the BSI system by testing neural decoders trained on uncued movement and imagery tasks. By developing a non-invasive BSI, tSCS can be timed to be delivered only during voluntary effort, which may have implications for improving rehabilitation.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: This study was conducted in accordance with the Declaration of Helsinki and has been approved by Washington University in St. Louis’ Institutional Review Board (IRB ID 202105168). All participants signed a written informed consent prior to the study and received financial compensation for their participation. Consent for publication: Not applicable. Competing interests: E.C.L. holds various patents in relation to the present work and is a founder and shareholder of Neurolutions, Inc., a company developing EEG technologies for stroke. All other authors declare no competing interests.
Figures







Update of
-
Development and evaluation of a non-invasive brain-spine interface using transcutaneous spinal cord stimulation.bioRxiv [Preprint]. 2024 Sep 19:2024.09.16.612897. doi: 10.1101/2024.09.16.612897. bioRxiv. 2024. Update in: J Neuroeng Rehabil. 2025 Apr 25;22(1):95. doi: 10.1186/s12984-025-01628-6. PMID: 39345398 Free PMC article. Updated. Preprint.
References
-
- Behrman AL, Bowden MG, Nair PM. Neuroplasticity after spinal cord injury and training: an emerging paradigm shift in rehabilitation and walking recovery. Phys Ther. 2006;86(10):1406–25. - PubMed
-
- Thuret S, Moon LDF, Gage FH. Therapeutic interventions after spinal cord injury. Nat Rev Neurosci. 2006;7(8):628–43. - PubMed
-
- Gill ML, Grahn PJ, Calvert JS, Linde MB, Lavrov IA, Strommen JA, et al. Neuromodulation of lumbosacral spinal networks enables independent stepping after complete paraplegia. Nat Med. 2018;24(11):1677–82. - PubMed
-
- Angeli CA, Boakye M, Morton RA, Vogt J, Benton K, Chen Y, et al. Recovery of Over-Ground walking after chronic motor complete spinal cord injury. N Engl J Med. 2018;379(13):1244–50. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

