Secretory Production of Plant Heme-Containing Globins by Recombinant Yeast via Precision Fermentation
- PMID: 40282823
- PMCID: PMC12026525
- DOI: 10.3390/foods14081422
Secretory Production of Plant Heme-Containing Globins by Recombinant Yeast via Precision Fermentation
Abstract
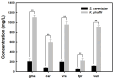
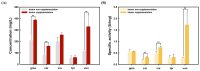
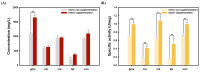
Leghemoglobin (LegHb) is a plant-derived heme-containing globin found in the root nodules of legumes like soybean that can be used as a food additive for red color and meaty flavor as a plant-based meat alternative. However, conventional extraction methods face challenges of low yield and high costs. To address this issue, precision fermentation with recombinant microorganisms has been applied for the sustainable large-scale production of plant leghemoglobins. This study attempted the production of plant legHbs using recombinant yeast strains, Saccharomyces cerevisiae and Komagatella phaffii. The plant legHb genes were identified from the genome of legumes such as soybean, chickpea, mung bean and overexpressed in yeast via extracellular secretion by the signal peptide and inducible promoters. Subsequently, hemin as a heme provider was added to the fermentation, resulting in increased levels of plant legHbs. In S. cerevisiae, gmaLegHb expression reached up to 398.1 mg/L, while in K. phaffii, gmaLegHb showed the highest production level, reaching up to 1652.7 mg/L. The secretory production of plant legHbs was further enhanced by replacing the signal peptide in the recombinant yeast. The secreted plant legHbs were purified by His-Tag from a culture supernatant or concentrated via precipitation using ammonium sulfate. These results suggest that the production of plant legHbs is significantly influenced by hemin and signal peptide. This study successfully demonstrates the production of the various plant legHbs other than soy legHb that can be used as natural colors and flavors for plant-based meat alternatives.
Keywords: Komagataella phaffii; Saccharomyces cerevisiae; plant leghemoglobin; precision fermentation; secretion.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Ritchie H., Rosado P., Roser M. Meat and dairy production. Our World in Data. 2019
-
- McLeod A. World Livestock 2011—Livestock in Food Security. Food and Agriculture Organization of the United Nations (FAO); Rome, Italy: 2011.
Grants and funding
LinkOut - more resources
Full Text Sources

