Aromatic Molecular Compatibility Attenuates Influenza Virus-Induced Acute Lung Injury via the Lung-Gut Axis and Lipid Droplet Modulation
- PMID: 40283905
- PMCID: PMC12030469
- DOI: 10.3390/ph18040468
Aromatic Molecular Compatibility Attenuates Influenza Virus-Induced Acute Lung Injury via the Lung-Gut Axis and Lipid Droplet Modulation
Abstract
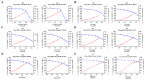
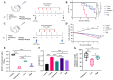
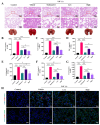
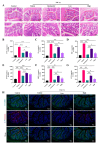
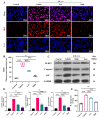
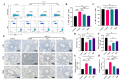
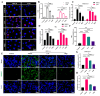
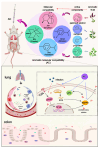
Background: Acute lung injury (ALI) is a major cause of death in patients with various viral pneumonias. Our team previously identified four volatile compounds from aromatic Chinese medicines. Based on molecular compatibility theory, we defined their combination as aromatic molecular compatibility (AC), though its therapeutic effects and underlying mechanisms remain unclear. Methods: This study used influenza A virus (IAV) A/PR/8/34 to construct cell and mouse models of ALI to explore AC's protective effects against viral infection. The therapeutic effect of AC was verified by evaluating the antiviral efficacy in the mouse models, including improvements in their lung and colon inflammation, oxidative stress, and the suppression of the NLRP3 inflammasome. In addition, 16S rDNA and lipid metabolomics were used to analyze the potential therapeutic mechanisms of AC. Results: Our in vitro and in vivo studies demonstrated that AC increased the survival of the IAV-infected cells and mice, inhibited influenza virus replication and the expression of proinflammatory factors in the lung tissues, and ameliorated barrier damage in the colonic tissues. In addition, AC inhibited the expression of ROS and the NLRP3 inflammasome and improved the inflammatory cell infiltration into the lung tissues. Finally, AC effectively regulated intestinal flora disorders and lipid metabolism in the model mice, significantly reduced cholesterol and triglyceride expression, and thus reduced the abnormal accumulation of lipid droplets (LDs) after IAV infection. Conclusions: In this study, we demonstrated that AC could treat IAV-induced ALIs through multiple pathways, including antiviral and anti-inflammatory pathways and modulation of the intestinal flora and the accumulation of LDs.
Keywords: NLRP3 inflammasome; acute lung injury; influenza A virus; lipid droplets; molecular compatibility; volatile compounds.
Conflict of interest statement
Author Zhongpeng Zhao was employed by Beijing Minhai Biotechnology Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Tangeretin attenuates acute lung injury in septic mice by inhibiting ROS-mediated NLRP3 inflammasome activation via regulating PLK1/AMPK/DRP1 signaling axis.Inflamm Res. 2024 Jan;73(1):47-63. doi: 10.1007/s00011-023-01819-8. Epub 2023 Dec 26. Inflamm Res. 2024. PMID: 38147126
-
Proanthocyanidins attenuates ferroptosis against influenza-induced acute lung injury in mice by reducing IFN-γ.Life Sci. 2023 Feb 1;314:121279. doi: 10.1016/j.lfs.2022.121279. Epub 2022 Dec 14. Life Sci. 2023. PMID: 36526043
-
Integrated network pharmacology and intestinal flora analysis to determine the protective effect of Xuanbai-Chengqi decoction on lung and gut injuries in influenza virus-infected mice.J Ethnopharmacol. 2022 Nov 15;298:115649. doi: 10.1016/j.jep.2022.115649. Epub 2022 Aug 17. J Ethnopharmacol. 2022. PMID: 35987410
-
Xijiao Dihuang decoction combined with Yinqiao powder promotes autophagy-dependent ROS decrease to inhibit ROS/NLRP3/pyroptosis regulation axis in influenza virus infection.Phytomedicine. 2024 Jun;128:155446. doi: 10.1016/j.phymed.2024.155446. Epub 2024 Feb 10. Phytomedicine. 2024. PMID: 38518643
-
Study on the activity and mechanism of herbal formula anti-infection powder (AIP) against influenza-virus-induced pneumonia through genetic susceptibility genes.J Ethnopharmacol. 2025 Jan 30;337(Pt 3):118959. doi: 10.1016/j.jep.2024.118959. Epub 2024 Oct 18. J Ethnopharmacol. 2025. PMID: 39426575
References
-
- Bao L., Deng W., Qi F., Lv Q., Song Z., Liu J., Gao H., Wei Q., Yu P., Xu Y., et al. Sequential infection with H1N1 and SARS-CoV-2 aggravated COVID-19 pathogenesis in a mammalian model, and co-vaccination as an effective method of prevention of COVID-19 and influenza. Signal Transduct. Target. Ther. 2021;6:200. doi: 10.1038/s41392-021-00618-z. - DOI - PMC - PubMed
-
- Yue H., Zhang M., Xing L., Wang K., Rao X., Liu H., Tian J., Zhou P., Deng Y., Shang J. The epidemiology and clinical characteristics of co-infection of SARS-CoV-2 and influenza viruses in patients during COVID-19 outbreak. J. Med. Virol. 2020;92:2870–2873. doi: 10.1002/jmv.26163. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

