α1 Adrenergic Receptors Mediate Panic-like Defensive Behavior in Alcohol-Drinking but Not Alcohol-Naïve Rats
- PMID: 40283921
- PMCID: PMC12030093
- DOI: 10.3390/ph18040484
α1 Adrenergic Receptors Mediate Panic-like Defensive Behavior in Alcohol-Drinking but Not Alcohol-Naïve Rats
Abstract
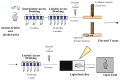
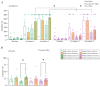
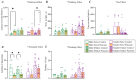
Background: Most animals display different defensive behavioral strategies during imminent or potential threats. These responses are relevant for understanding human behavioral disorders. In addition, α1 adrenergic receptors (α1ARs) are blocked by prazosin and regulate a diverse set of behaviors, including alcohol drinking related to anxiety in humans, alcohol intake in rats, responses to strong acute stresses (like restraint), and several forms of cognitive flexibility. However, the role of α1ARs in regulating panic-like escape behavior remains unexplored. Methods: Male and female Wistar rats were chronic alcohol drinkers and age-matched alcohol naïves. Animals received an injection of 0.75 mg/kg of prazosin or vehicle and then were exposed to the elevated T maze (ETM) to evaluate avoidance and escape behavior. One week later, animals underwent the light-dark test (LDT) and open field test. Results: α1AR inhibition with prazosin increased latency for escape in male and female alcohol drinkers, with no significant effects in alcohol-naïve controls. There were also interesting impacts from alcohol drinking, including a decrease in ETM avoidance in female but not male drinkers. In addition, prazosin increased latency to enter the dark in LDT in female drinkers and male naïves. Although prazosin also decreased the number of transitions in males, no differences were found in open-field locomotion. Conclusions: These results suggest that α1ARs mediate escape-like behavior in male and female alcohol drinkers, shedding light on a novel therapy for alcohol problems related to panic and anxiety.
Keywords: alcohol drinking; anxiety; panic; α1 adrenergic receptors.
Conflict of interest statement
The author declare no conflict of interest.
Figures
References
Grants and funding
LinkOut - more resources
Full Text Sources

