New Cannabinoids and Chlorin-Type Metabolites from the Flowers of Cannabis sativa L.: A Study on Their Neuroblastoma Activity
- PMID: 40283956
- PMCID: PMC12030031
- DOI: 10.3390/ph18040521
New Cannabinoids and Chlorin-Type Metabolites from the Flowers of Cannabis sativa L.: A Study on Their Neuroblastoma Activity
Abstract
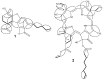
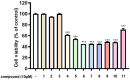
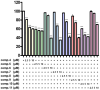
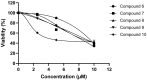
Background/Objectives: Cannabis sativa has been utilized for medical purposes for thousands of years. It continues to be recognized as a plant with an extensive variety of medicinal and nutraceutical uses today. In this study, a chemical investigation of the flowers of C. sativa isolated by using a variety of chromatographic techniques led to the isolation of eleven compounds. These purified compounds were evaluated for antitumor activity against SK-N-SH neuroblastoma cells. Methods: The compounds were isolated by using chromatographic techniques. Their structures were identified by the examination of spectroscopic methods, including 1D (1H, 13C, and DEPT) and 2D (COSY, HSQC, HMBC, and NOESY) nuclear magnetic resonance (NMR) spectra and mass spectrum, together with the comparison to those reported previously in the literature. The evaluation of toxicity on SK-N-SH cells was performed by the MTT method. Results: Eleven compounds were isolated from the flowers of C. sativa, including two new compounds, namely cannabielsoxa (1), 132-hydroxypheophorbide c ethyl ester (2), and six known cannabinoids (6-11), together with the first isolation of chlorin-type compounds: pyropheophorbide A (3), 132-hydroxypheophorbide b ethyl ester (4), and ligulariaphytin A (5) from this plant. The results also demonstrated that cannabinoid compounds had stronger inhibitory effects on neuroblastoma cells than chlorin-type compounds. Conclusions: The evaluation of the biological activities of compounds showed that compounds 4-10 could be considered as the potential compounds for antitumor effects against neuroblastomas. This is also highlighted by using docking analysis. Additionally, the results of this study also suggest that these compounds have the potential to be developed into antineuroblastoma products.
Keywords: Cannabis sativa L.; SK-N-SH cells; cannabinoid; chlorin-type compound.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
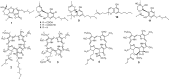
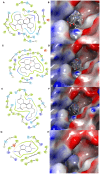
 ) and HMBC (
) and HMBC ( ) correlations of 1 and 2.
) correlations of 1 and 2.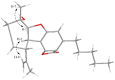

Similar articles
-
NMR assignments of the major cannabinoids and cannabiflavonoids isolated from flowers of Cannabis sativa.Phytochem Anal. 2004 Nov-Dec;15(6):345-54. doi: 10.1002/pca.787. Phytochem Anal. 2004. PMID: 15595449
-
Simultaneous quantification of terpenes and cannabinoids by reversed-phase LC-APCI-MS/MS in Cannabis sativa L. samples combined with a subsequent chemometric analysis.Anal Bioanal Chem. 2024 Jul;416(18):4193-4206. doi: 10.1007/s00216-024-05349-y. Epub 2024 May 25. Anal Bioanal Chem. 2024. PMID: 38795214 Free PMC article.
-
The Medicinal Natural Products of Cannabis sativa Linn.: A Review.Molecules. 2022 Mar 4;27(5):1689. doi: 10.3390/molecules27051689. Molecules. 2022. PMID: 35268790 Free PMC article. Review.
-
Monitoring Metabolite Profiles of Cannabis sativa L. Trichomes during Flowering Period Using 1H NMR-Based Metabolomics and Real-Time PCR.Planta Med. 2016 Aug;82(13):1217-23. doi: 10.1055/s-0042-108058. Epub 2016 Jun 23. Planta Med. 2016. PMID: 27336318
-
Non-Cannabinoid Metabolites of Cannabis sativa L. with Therapeutic Potential.Plants (Basel). 2021 Feb 20;10(2):400. doi: 10.3390/plants10020400. Plants (Basel). 2021. PMID: 33672441 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

