The Impact of Doxycycline as an Adjunctive Therapy on Prostate-Specific Antigen, Quality of Life, and Cognitive Function in Metastatic Prostate Cancer Patients: A Phase II Randomized Controlled Trial
- PMID: 40284400
- PMCID: PMC12030710
- DOI: 10.3390/pharmaceutics17040404
The Impact of Doxycycline as an Adjunctive Therapy on Prostate-Specific Antigen, Quality of Life, and Cognitive Function in Metastatic Prostate Cancer Patients: A Phase II Randomized Controlled Trial
Abstract
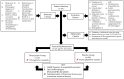
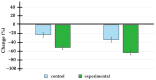
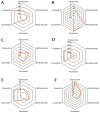
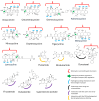
Background/Objectives: Metastatic prostate cancer remains a major clinical challenge, with limited therapeutic options. Doxycycline, a tetracycline antibiotic with anti-inflammatory properties, has shown potential as an adjunctive therapy. This study aimed to evaluate its efficacy in reducing prostate-specific antigen (PSA) levels and improving quality of life in patients receiving standard treatment for metastatic prostate cancer. Methods: This phase II, double-blind, randomized controlled trial included 45 participants (aged 57-81 years) assigned to doxycycline (100 mg daily) or a placebo for six months. The primary outcome was the percentage change in PSA levels at 3 and 6 months. Secondary outcomes included quality of life (EQ-5D-5L), cognitive function (Mini-Mental State Examination), and glucose levels. Additionally, a structure-activity relationship (SAR) analysis was performed through an extensive bibliographic review to identify pharmacophores responsible for doxycycline's biological activity, particularly its tetracyclic core. The SAR analysis included tetracyclines and derivatives, androgen-targeting agents, and other pharmacologically relevant molecules used in prostate cancer therapy. Statistical analysis was conducted using multivariate logistic regression. Results: At six months, the doxycycline group showed a median PSA reduction of 60% compared to 10% in the placebo group (p = 0.043). A ≥50% reduction in PSA levels was observed in 71.4% of patients receiving doxycycline versus 20.8% in the placebo group (p = 0.001), with an adjusted relative risk of 10.309 (95% CI: 2.359-45.055, p = 0.002). Quality of life improved, with 7.1% of doxycycline-treated patients reporting poor quality of life compared to 42.9% in the placebo group (p = 0.028). A slight improvement in cognitive function was also noted (p = 0.037). SAR analysis suggested that the tetracyclic ring of doxycycline may play a crucial role in its observed biological effects. Conclusions: Doxycycline demonstrates potential as an adjunctive therapy in metastatic prostate cancer by reducing PSA levels and improving quality of life. The SAR analysis supports the hypothesis that its tetracyclic structure may be responsible for its therapeutic effects. Further large-scale trials are warranted to confirm these findings.
Keywords: doxycycline; metastatic prostate cancer; prostate-specific antigen (PSA); quality of life; randomized controlled trial; structure–activity relationship (SAR).
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
-
- Guzman-Esquivel J., Murillo-Zamora E., Ortiz-Mesina M., Galvan-Salazar H.R., De-Leon-Zaragoza L., Casarez-Price J.C., Delgado-Enciso J., Delgado-Enciso I. Regional and National Burden of Prostate Cancer: Incidence, Mortality, Years of Life Lost, and Disability-Adjusted Life Years, in Mexico and Latin America from 1990 to 2019. Int. Urol. Nephrol. 2023;55:2155–2160. doi: 10.1007/s11255-023-03653-7. - DOI - PMC - PubMed
-
- Bikbov B., Purcell C.A., Levey A.S., Smith M., Abdoli A., Abebe M., Adebayo O.M., Afarideh M., Agarwal S.K., Agudelo-Botero M., et al. Global, Regional, and National Burden of Chronic Kidney Disease, 1990–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet. 2020;395:709–733. doi: 10.1016/S0140-6736(20)30045-3. - DOI - PMC - PubMed
-
- Jimenez-Rios M.A., Scavuzzo A., Noverón N.R., García-Arango C., Calvo-Vazquez I., Hurtado-Vázquez A., Arrieta-Rodriguez O.G., Davila-Jimenez M.A., Sighinolfi M.C., Rocco B. Lethal Prostate Cancer in Mexico: Data from the Can.Prost Mexican Registry and a Project for Early Detection. Cancers. 2024;16:3675. doi: 10.3390/cancers16213675. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

