Delivery of PLGA-Loaded Influenza Vaccine Microparticles Using Dissolving Microneedles Induces a Robust Immune Response
- PMID: 40284505
- PMCID: PMC12030082
- DOI: 10.3390/pharmaceutics17040510
Delivery of PLGA-Loaded Influenza Vaccine Microparticles Using Dissolving Microneedles Induces a Robust Immune Response
Abstract
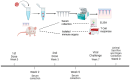
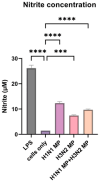
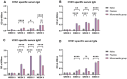
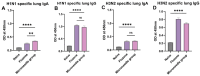
Background: Influenza virus is one of the major respiratory virus infections that is a global health concern. Although there are already approved vaccines, most are administered via the intramuscular route, which is usually painful, leading to vaccine hesitancy. To this end, exploring the non-invasive, transdermal vaccination route using dissolving microneedles would significantly improve vaccine compliance. Research on innovative vaccine delivery systems, such as antigen-loaded PLGA microparticles, has the potential to pave the way for a broader range of vaccine candidates. Methods: In this proof-of-concept study, a combination of the inactivated influenza A H1N1 virus and inactivated influenza A H3N2 virus were encapsulated in a biodegradable poly (lactic-co-glycolic acid) (PLGA) polymeric matrix within microparticles, which enhanced antigen presentation. The antigen PLGA microparticles were prepared separately using a double emulsion (w/o/w), lyophilized, and characterized. Next, the vaccine microparticles were assessed in vitro in dendritic cells (DC 2.4) for immunogenicity. To explore pain-free transdermal vaccination, the vaccine microparticles were loaded into dissolving microneedles and administered in mice (n = 5). Results: Our vaccination study demonstrated that the microneedle-based vaccine elicited strong humoral responses as demonstrated by high antigen-specific IgA, IgG, IgG1, and IgG2a antibodies in serum samples and IgA in lung supernatant. Further, the vaccine also elicited a strong cellular response as evidenced by high levels of CD4+ and CD8a+ T cells in lymphoid organs such as the lymph nodes and spleen. Conclusion: The delivery of influenza vaccine-loaded PLGA microparticles using microneedles would be beneficial to individuals experiencing needle-phobia, as well as the geriatric and pediatric population.
Keywords: PLGA; influenza; microneedles; microparticles; polymer.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
Vaccine-Induced Immunity Elicited by Microneedle Delivery of Influenza Ectodomain Matrix Protein 2 Virus-like Particle (M2e VLP)-Loaded PLGA Nanoparticles.Int J Mol Sci. 2023 Jun 25;24(13):10612. doi: 10.3390/ijms241310612. Int J Mol Sci. 2023. PMID: 37445784 Free PMC article.
-
Adjuvanted-SARS-CoV-2 Spike Protein-Based Microparticulate Vaccine Delivered by Dissolving Microneedles Induces Humoral, Mucosal, and Cellular Immune Responses in Mice.Pharmaceuticals (Basel). 2023 Aug 10;16(8):1131. doi: 10.3390/ph16081131. Pharmaceuticals (Basel). 2023. PMID: 37631046 Free PMC article.
-
Sustained release of inactivated H1N1 virus from degradable microparticles for extended vaccination.Eur J Pharm Biopharm. 2024 Sep;202:114388. doi: 10.1016/j.ejpb.2024.114388. Epub 2024 Jun 28. Eur J Pharm Biopharm. 2024. PMID: 38945409
-
Dissolving Microneedles Loaded with Nanoparticle Formulation of Respiratory Syncytial Virus Fusion Protein Virus-like Particles (F-VLPs) Elicits Cellular and Humoral Immune Responses.Vaccines (Basel). 2023 Apr 18;11(4):866. doi: 10.3390/vaccines11040866. Vaccines (Basel). 2023. PMID: 37112778 Free PMC article.
-
Microneedles: A New Generation Vaccine Delivery System.Micromachines (Basel). 2021 Apr 14;12(4):435. doi: 10.3390/mi12040435. Micromachines (Basel). 2021. PMID: 33919925 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

