Evolution of Epstein-Barr Virus Infection Seroprevalence in a French University Hospital over 11 Years, Including the COVID-19 Pandemic, 2013-2023
- PMID: 40284570
- PMCID: PMC12029913
- DOI: 10.3390/microorganisms13040733
Evolution of Epstein-Barr Virus Infection Seroprevalence in a French University Hospital over 11 Years, Including the COVID-19 Pandemic, 2013-2023
Abstract
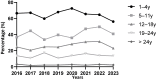
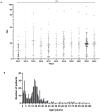
Epstein-Barr virus (EBV) is one of the most prevalent human viruses worldwide. The COVID-19 pandemic, with its social distancing measures, has disrupted the circulation of many viruses. Delayed EBV primary infection is known to increase the risk of secondary conditions, including infectious mononucleosis, multiple sclerosis, and Hodgkin's lymphoma. In this context, we aimed to investigate whether EBV seroprevalence has been affected over time, particularly in relation to the COVID-19 period, by analyzing all patients admitted to Amiens University Hospital from January 2013 to December 2023 who underwent EBV serology. During this period, 19,771 EBV serologies were performed and analyzed. The total seropositive rate of EBV infections approached 90%, considering all non-negative serological profiles, with the rate stabilizing after 2017. The number of EBV serologies increased significantly until 2016, as well as the age of the screened patients. Less than 3% of patients remain seronegative after 25 years, indicating a seroprevalence of around 97%. The overall primary infection rate was 2.6%. There was no significant difference in the number of primary infections in 2020-2021, the years associated with confinements and curfews in France in the context of the COVID-19 pandemic, compared with the other years. The overall EBV seroprevalence and age of primary infection remained stable during the study period, suggesting a moderate impact of the COVID-19 pandemic on seroprevalence in this cohort.
Keywords: COVID; EBV; France; primary infection; seroprevalence.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



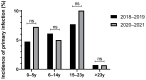
References
-
- Hjalgrim H., Smedby K.E., Rostgaard K., Molin D., Hamilton-Dutoit S., Chang E.T., Ralfkiaer E., Sundström C., Adami H.-O., Glimelius B., et al. Infectious Mononucleosis, Childhood Social Environment, and Risk of Hodgkin Lymphoma. Cancer Res. 2007;67:2382–2388. doi: 10.1158/0008-5472.CAN-06-3566. - DOI - PubMed
LinkOut - more resources
Full Text Sources

