The Vaginally Exposed Extracellular Vesicle of Gardnerella vaginalis Induces RANK/RANKL-Involved Systemic Inflammation in Mice
- PMID: 40284791
- PMCID: PMC12029968
- DOI: 10.3390/microorganisms13040955
The Vaginally Exposed Extracellular Vesicle of Gardnerella vaginalis Induces RANK/RANKL-Involved Systemic Inflammation in Mice
Abstract
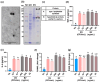
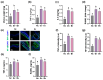
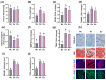
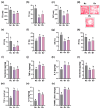
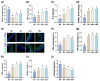
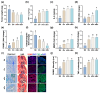
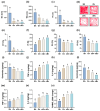
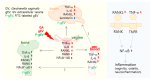
Gardnerella vaginalis (GV), an opportunistic pathogen excessively proliferated in vaginal dysbiosis, causes systemic inflammation including vaginitis, neuroinflammation, and osteitis. To understand its systemic inflammation-triggering factor, we purified extracellular vesicles isolated from GV (gEVs) and examined their effect on the occurrence of vaginitis, osteitis, and neuroinflammation in mice with and without ovariectomy (Ov). The gEVs consisted of lipopolysaccharide, proteins, and nucleic acid and induced TNF-α and RANKL expression in macrophage cells. When the gEVs were vaginally exposed in mice without Ov, they significantly induced RANK, RANKL, and TNF-α expression and NF-κB+ cell numbers in the vagina, femur, hypothalamus, and hippocampus, as observed in GV infection. The gEVs decreased time spent in the open field (OT) in the elevated plus maze test by 47.3%, as well as the distance traveled in the central area (DC) by 28.6%. In the open field test, they also decreased the time spent in the central area (TC) by 39.3%. Additionally, gEVs decreased spontaneous alteration (SA) in the Y-maze test by 33.8% and the recognition index (RI) in the novel object recognition test by 26.5%, while increasing the immobility time (IT) in the tail suspension test by 36.7%. In mice with OV (Ov), the gEVs also induced RANK, RANKL, and TNF-α expression and increased NF-κB+ cell numbers in the vagina, femur, hypothalamus, and hippocampus compared to vehicle-treated mice. When gEVs were exposed to mice with Ov, gEVs also reduced the DC, TC, OT, SA, and RI to 62.1%, 62.7%, 28.2%, 90.7%, and 85.4% of mice with Ov, respectively, and increased IT to 122.9% of mice with Ov. Vaginally exposed fluorescein-isothiocyanate-tagged gEVs were detected in the blood, femur, and hippocampus. These findings indicate that GV-derived gEVs may induce systemic inflammation through the activation of RANK/RANKL-involved NF-κB signaling, leading to systemic disorders including vaginitis, osteoporosis, depression, and cognitive impairment. Therefore, gEVs may be an important risk factor for vaginitis, osteoporosis, depression, and cognitive impairment in women.
Keywords: Gardnerella vaginalis; extracellular vesicle; osteoporosis; psychiatric disorder; vaginitis.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
Grants and funding
LinkOut - more resources
Full Text Sources

