Experimental Infection of Rats with Influenza A Viruses: Implications for Murine Rodents in Influenza A Virus Ecology
- PMID: 40284938
- PMCID: PMC12030792
- DOI: 10.3390/v17040495
Experimental Infection of Rats with Influenza A Viruses: Implications for Murine Rodents in Influenza A Virus Ecology
Abstract
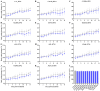
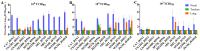
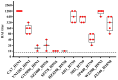
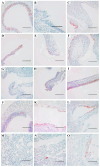
Rattus norvegicus (brown rat), a widely distributed rodent and common biomedical model, is a known reservoir for many zoonotic pathogens but has not been traditionally recognized as a host for influenza A virus (IAV). To evaluate their susceptibility, we intranasally inoculated Sprague-Dawley rats with various IAV subtypes, including H5Nx, H7N9, H9N2, H10N8 and the 2009 pandemic H1N1. All strains productively infected the rats, inducing seroconversion without overt clinical signs. While replication efficiency varied, all viruses caused significant lung injury with a preferential tropism for the upper respiratory tract. Investigation of receptor distribution revealed a predominance of α2,3-linked sialic acid (SA) in the nasal turbinates and trachea, whereas α2,6-linked SA was more abundant in the lungs. Notably, both receptor types coexisted throughout the respiratory tract, aligning with the observed tissue-specific replication patterns and broad viral infectivity. These findings demonstrate that rats are permissive hosts for multiple IAV subtypes, challenging their exclusion from IAV ecology. The asymptomatic yet pathogenic nature of infection, combined with the global synanthropy of rats, underscores their potential role as cryptic reservoirs in viral maintenance and transmission. This study highlights the need for expanded surveillance of rodents in influenza ecology to mitigate zoonotic risks.
Keywords: host range; interspecies transmission; sialic acid receptor; surveillance; zoonotic influenza.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- World Health Organization (WHO) Global Influenza Programme. [(accessed on 27 February 2025)]. Available online: https://www.who.int/teams/global-influenza-programme/avian-influenza/mon....
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

