Indocyanine Green-Loaded Quenched Nanoliposomes as Activatable Theranostics for Cancer
- PMID: 40286013
- PMCID: PMC11990451
- DOI: 10.3390/molecules30071414
Indocyanine Green-Loaded Quenched Nanoliposomes as Activatable Theranostics for Cancer
Abstract
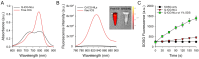
Photodynamic therapy (PDT) and photothermal therapy (PTT) are considered to be one of the most effective methods for treating cancer due to their noninvasive nature, high effectiveness, and fewer side effects compared to standard therapeutic modalities for cancer. However, conventional always-on types of PDT and PTT agents have basic drawbacks in their in vivo applications, which include the unwanted generation of strong fluorescence signals and phototoxicity in normal tissues, including blood vessels, when exposed to light, resulting in poor imaging contrast and unwanted phototoxicity. Here, we propose indocyanine green-loaded quenched nanoliposomes (Q-ICG-NLs) as an activatable nanotheranostics. Q-ICG-NLs showed significant quenching in near-infrared fluorescence emission and singlet oxygen generation upon light irradiation. The photothermal effect of Q-ICG-NLs was 1.3 times greater than free indocyanine green. Its fluorescence and singlet oxygen generation were largely restored when taken up into cancer cells, enabling the selective detection and phototherapy of cancer cells. These results suggest that Q-ICG-NLs can be effectively used for selective near-infrared fluorescence imaging and the subsequent image-guided PDT and PTT of cancers.
Keywords: activatable; indocyanine green; nanoliposome; phototherapy.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





Similar articles
-
Perfluorooctyl bromide & indocyanine green co-loaded nanoliposomes for enhanced multimodal imaging-guided phototherapy.Biomaterials. 2018 May;165:1-13. doi: 10.1016/j.biomaterials.2018.02.041. Epub 2018 Feb 24. Biomaterials. 2018. PMID: 29500978
-
Tumor Cell-Targeting and Tumor Microenvironment-Responsive Nanoplatforms for the Multimodal Imaging-Guided Photodynamic/Photothermal/Chemodynamic Treatment of Cervical Cancer.Int J Nanomedicine. 2024 Jun 13;19:5837-5858. doi: 10.2147/IJN.S466042. eCollection 2024. Int J Nanomedicine. 2024. PMID: 38887692 Free PMC article.
-
Low Power Single Laser Activated Synergistic Cancer Phototherapy Using Photosensitizer Functionalized Dual Plasmonic Photothermal Nanoagents.ACS Nano. 2019 Feb 26;13(2):2544-2557. doi: 10.1021/acsnano.8b09552. Epub 2019 Feb 11. ACS Nano. 2019. PMID: 30730695
-
Indocyanine green-based nanomedicine for theranostic applications.Biomater Sci. 2025 Aug 5;13(16):4321-4357. doi: 10.1039/d5bm00670h. Biomater Sci. 2025. PMID: 40653875 Review.
-
Tailoring Indocyanine Green J-Aggregates for Imaging, Cancer Phototherapy, and Drug Delivery: A Review.ACS Appl Bio Mater. 2024 Aug 19;7(8):5121-5135. doi: 10.1021/acsabm.4c00651. Epub 2024 Jul 23. ACS Appl Bio Mater. 2024. PMID: 39039943 Review.
References
-
- Kim H.J., Lee S.S., Sohn D.K., Yoon H.M., Park K.L., Park S.J., Choi Y. Multiporous PMMA microballs as a novel fluorescence tissue marker. Chem. Eng. J. 2024;496:154267. doi: 10.1016/j.cej.2024.154267. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

