Cholinium-Based Ionic Liquids Modulate Protein Stability: A Comparative Study of Enzymes and Albumins
- PMID: 40286162
- PMCID: PMC11990322
- DOI: 10.3390/molecules30071574
Cholinium-Based Ionic Liquids Modulate Protein Stability: A Comparative Study of Enzymes and Albumins
Abstract
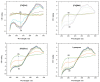
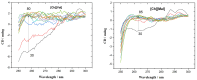
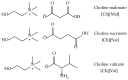
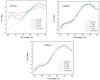
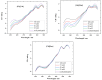
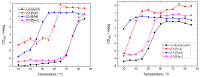
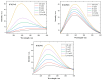
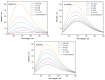
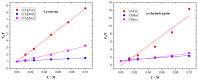
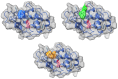
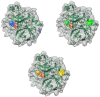
This work aims to assess the intermolecular interaction of choline ionic liquids (ILs) (choline malonate ([Ch][Mal]), choline succinate ([Ch][Suc]), and choline valinate ([Ch][Val]) with two enzymes (lysozyme and α-chymotrypsin). We evaluated the state of the tertiary protein structure using circular dichroism (CD) spectrometry and quantified the binding parameters of the binding of the ionic liquids to the enzymes by fluorescence spectroscopy. The binding energies of the enzymes and the localization of ions on them were estimated using the molecular docking. We then analyzed the relationship between the enzymes' thermostability and their tendency towards aggregation in the enzyme/ionic liquid systems. The obtained results were compared with previous data on albumins to identify similarities and differences between the behavior of enzymes and albumins in ionic liquid solutions. Despite the comparable values of the binding constants, the effect of ionic liquids on the thermostability of enzymes was the opposite of their effect on albumins. In addition, although these ionic liquids promoted aggregation in both enzymes and albumins, this effect was much more pronounced for albumins.
Keywords: binding; enzyme; ionic liquid; protein; structure.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Bisht M., Mondal D., Pereira M.M., Freire M.G., Venkatesu P., Coutinho J.A.P. Long-term protein packaging in cholinium-based ionic liquids: Improved catalytic activity and enhanced stability of cytochrome c against multiple stresses. Green Chem. 2017;19:4900–4911. doi: 10.1039/C7GC02011B. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

