Evaluating the Influence of Different Serum-Free Culture Conditions on the Production and Function of Natural Killer Cell-Derived Extracellular Vesicles
- PMID: 40292387
- PMCID: PMC12025879
- DOI: 10.1002/jex2.70049
Evaluating the Influence of Different Serum-Free Culture Conditions on the Production and Function of Natural Killer Cell-Derived Extracellular Vesicles
Abstract
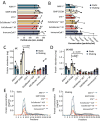
Natural killer (NK) cells are exploited in cellular therapies for cancer. While NK cell therapies are efficient against haematological cancers, it has been difficult to target solid tumours due to low tumour infiltration and a hostile tumour microenvironment. NK-cell derived extracellular vesicles (NK-EVs) target and kill cancer cells in vitro and represent an alternative treatment strategy for solid tumours. To exploit their potential, it is necessary to standardize NK-EV production protocols. Here, we have performed a comparative analysis of EVs from the human NK-92 cell line cultured in five serum-free commercial media optimized for growth of human NK cells and one serum-free medium for growth of lymphocytes. The effect of growing the NK-92 cells in static cell cultures versus shaking flasks was compared. EVs were purified via ultracentrifugation followed by size-exclusion chromatography. We found that there were no significant differences in EV yield from NK-92 cells grown under static or dynamic conditions. However, we found clear differences between the different culture media in terms of EV purity as assessed by the enrichment of the CD63 and CD81 markers in the isolates that translated into their capacity to induce apoptosis of the colon cancer cell line HCT 116. These findings will be instructive for the design of future production protocols for therapeutic NK-cell derived EVs.
Keywords: NK‐92 cells; cancer; extracellular vesicles; serum‐free; therapy.
© 2025 The Author(s). Journal of Extracellular Biology published by Wiley Periodicals LLC on behalf of International Society for Extracellular Vesicles.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
-
- Aarsund, M. , Segers F. M., Wu Y., and Inngjerdingen M.. 2022. “Comparison of Characteristics and Tumor Targeting Properties of Extracellular Vesicles Derived From Primary NK Cells or NK‐Cell Lines Stimulated With IL‐15 or IL‐12/15/18 .” Cancer Immunology, Immunotherapy 71, no. 9: 2227–2238. - PMC - PubMed
-
- Borrelli, D. A. , Yankson K., Shukla N., Vilanilam G., Ticer T., and Wolfram J.. 2018. “Extracellular Vesicle Therapeutics for Liver Disease.” Journal of Controlled Release 273: 86–98. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
