Methotrexate Enhances Atherosclerosis Progression via Impairment of Folate Pathway in a Microminipig Model
- PMID: 40295005
- PMCID: PMC12041998
- DOI: 10.21873/invivo.13930
Methotrexate Enhances Atherosclerosis Progression via Impairment of Folate Pathway in a Microminipig Model
Abstract
Background/aim: As the pathophysiology of Microminipigs (μMPs) is similar to that of human, μMPs are useful in atherosclerosis research. To clarify the effect of methotrexate (MTX) on atherosclerosis, we investigated the pathology of MTX-induced atherosclerosis lesion exacerbation in μMPs fed a high-fat and high-cholesterol diet (HFHCD).
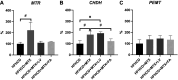
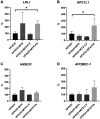
Materials and methods: The μMPs were divided into four groups: HFHCD, HFHCD+MTX, HFHCD+MTX+leucovorin (LV), and HFHCD+MTX+folic acid (FA), and fed for two weeks. Laboratory tests including blood lipid, FA, and homocysteine (Hcy) levels, and pathological evaluation of the atherosclerosis lesion area and thickness were performed. Hepatic and jejunal gene expressions related to lipid and folate metabolism pathways including 5-methyltetrahydrofolate-homocysteine methyltransferase (MTR) were monitored using RT-PCR.
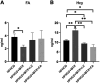
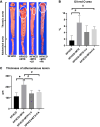
Results: The HFHCD+MTX group showed increased blood Hcy (p<0.01) and decreased FA levels (p<0.05) in accordance with increased hepatic MTR mRNA expression (p<0.1) and exacerbation of atherosclerosis (p=0.051 for lesion area and p=0.045 for lesion thickness) compared to the HFHCD group. Administration of LV or FA attenuated the MTX-induced increase in the Hcy level (p<0.01), atherosclerosis lesion thickness (p<0.1), and MTR mRNA expression (p<0.1 in HFHCD+MTX vs. HFHCD+MTX+LV groups).
Conclusion: MTX exacerbated HFHCD-induced atherosclerosis mediated through reduced blood FA and the subsequent increase of Hcy in μMPs, indicating that the μMP model may advance cardio-oncology research by providing useful experimental approaches. As MTX is administered for rheumatoid arthritis and malignant tumors in humans, atherosclerosis exacerbation should be acknowledged as a possible adverse effect of MTX treatment.
Keywords: Atherosclerosis; animal model; folic acid; high-fat and high-cholesterol diet; hyperhomocysteinemia; leucovorin; methotrexate; microminipig.
Copyright © 2025, International Institute of Anticancer Research (Dr. George J. Delinasios), All rights reserved.
Conflict of interest statement
The Authors declare that no conflicts of interest exist.
Figures


Similar articles
-
Association of hyperhomocysteinemia with genetic variants in key enzymes of homocysteine metabolism and methotrexate toxicity in rheumatoid arthritis patients.Inflamm Res. 2018 Aug;67(8):703-710. doi: 10.1007/s00011-018-1161-8. Epub 2018 May 23. Inflamm Res. 2018. PMID: 29796841
-
Choline deficiency and methotrexate treatment induces marked but reversible changes in hepatic folate concentrations, serum homocysteine and DNA methylation rates in rats.J Am Coll Nutr. 1995 Oct;14(5):480-5. doi: 10.1080/07315724.1995.10718539. J Am Coll Nutr. 1995. PMID: 8522727
-
Homocysteine and folate status in methotrexate-treated patients with rheumatoid arthritis.Rheumatology (Oxford). 2002 Jun;41(6):658-65. doi: 10.1093/rheumatology/41.6.658. Rheumatology (Oxford). 2002. PMID: 12048292 Clinical Trial.
-
Folate supplementation during methotrexate therapy for rheumatoid arthritis.Clin Exp Rheumatol. 2010 Sep-Oct;28(5 Suppl 61):S102-9. Epub 2010 Oct 28. Clin Exp Rheumatol. 2010. PMID: 21044441 Review.
-
Folate supplementation and methotrexate treatment in rheumatoid arthritis: a review.Rheumatology (Oxford). 2004 Mar;43(3):267-71. doi: 10.1093/rheumatology/keh088. Epub 2004 Jan 6. Rheumatology (Oxford). 2004. PMID: 14963199 Review.
References
-
- The top 10 causes of death, World Health Organisation, 2020. Available at: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death. [Last accessed on May 4, 2024]
-
- Okamura T, Tsukamoto K, Arai H, Fujioka Y, Ishigaki Y, Koba S, Ohmura H, Shoji T, Yokote K, Yoshida H, Yoshida M, Deguchi J, Dobashi K, Fujiyoshi A, Hamaguchi H, Hara M, Harada-Shiba M, Hirata T, Iida M, Ikeda Y, Ishibashi S, Kanda H, Kihara S, Kitagawa K, Kodama S, Koseki M, Maezawa Y, Masuda D, Miida T, Miyamoto Y, Nishimura R, Node K, Noguchi M, Ohishi M, Saito I, Sawada S, Sone H, Takemoto M, Wakatsuki A, Yanai H. Japan Atherosclerosis Society (JAS) guidelines for prevention of atherosclerotic cardiovascular diseases 2022. J Atheroscler Thromb. 2024;31(6):641–853. doi: 10.5551/jat.GL2022. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
