Sexual dimorphism in benign adrenocortical tumours
- PMID: 40296186
- PMCID: PMC12068951
- DOI: 10.1093/ejendo/lvaf088
Sexual dimorphism in benign adrenocortical tumours
Abstract
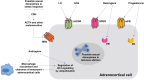
Benign adrenocortical tumours are the most common adrenal neoplasms. Evidence over the past few decades has highlighted sex differences in their prevalence, clinical characteristics, and treatment outcomes. Cortisol-producing adenomas causing either Cushing's syndrome, particularly those with PRKACA or GNAS somatic mutations associated with a more severe phenotype, or mild autonomous cortisol secretion (MACS) are more commonly observed in women. The mechanisms underpinning this sexual dimorphism remain to be fully elucidated. Studies in mice have revealed a protective role of androgens in males, leading to a decelerated growth rate of adrenocortical cells. Furthermore, evidence from human adrenal tumour tissue suggests that oestrogen, progesterone, and luteinising hormone/choriogonadotropin signalling in the adrenal cortex may play a role in adrenal tumourigenesis and steroid production. Clinically, this is supported by the increased incidence of cortisol-producing adrenocortical adenomas or nodular hyperplasia during puberty, pregnancy, and menopause. Notably, women with MACS seem to be more vulnerable to the harmful effects of cortisol excess and carry a higher mortality risk than men. Women with aldosterone-producing adenomas have a higher prevalence of somatic KCNJ5 mutations than men, and patients harbouring these mutations are likely to have more favourable clinical outcomes after adrenalectomy. In this review, we summarise the possible mechanisms behind the sexual dimorphism of benign adrenocortical tumours and provide an up-to-date overview of the sex-specific differences in their prevalence, clinical presentation, and outcomes, focusing on cortisol and aldosterone excess. Considering sexual dimorphism is crucial to guide diagnosis and management, and to counsel these patients for optimised care.
Keywords: Cushing's syndrome; adrenal tumour; mild autonomous cortisol secretion; primary aldosteronism; sexual dimorphism.
© The Author(s) 2025. Published by Oxford University Press on behalf of European Society of Endocrinology.
Conflict of interest statement
Conflict of interest: None declared.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

