Effect on Pain Following One Session of Botulinum Toxin Type A in Patients With Jaw Myalgia: A Randomised Double-Blind Controlled Multicentre Pilot Study
- PMID: 40296202
- PMCID: PMC12037931
- DOI: 10.1111/joor.13915
Effect on Pain Following One Session of Botulinum Toxin Type A in Patients With Jaw Myalgia: A Randomised Double-Blind Controlled Multicentre Pilot Study
Abstract
Background: Botulinum toxin Type A (BTX-A) is increasingly used in the management of myofascial pain; however, the evidence to support this treatment modality is still inconclusive.
Objective: To evaluate the change in days with functional jaw pain after one session of BTX-A or placebo injections into the masseter and temporalis muscles in subjects with jaw myalgia. The hypothesis was that BTX-A is effective in reducing the number of days with functional jaw pain.
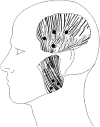
Methods: This was a randomised, double-blind, parallel group study in subjects with jaw myalgia. After randomisation, BTX-A or placebo injections were made in the masseter and temporalis muscles. The number of days with jaw pain on function was evaluated after 2 months. Adverse events were registered.
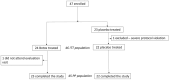
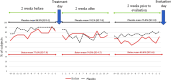
Results: Twenty-three subjects were randomly assigned to BTX-A and 22 to placebo. Between baseline and the 2-month follow-up, the number of days with jaw pain at function changed from a median (interquartile range) of 14 (4) to 10.5 (6) days in the BTX-A group and from 14.0 (1.5) to 14 (5) in the saline group (p < 0.585). Adverse events were mild and transient and similar in the two groups.
Conclusion: A single treatment of BTX-A injections in the masseter and temporalis muscles was not effective in treating jaw myalgia. However, an adequately powered study might provide sufficient support for efficacy.
Keywords: botulinum toxin type A; myofascial pain; orofacial pain; temporomandibular disorders; treatment.
© 2024 The Author(s). Journal of Oral Rehabilitation published by John Wiley & Sons Ltd.
Conflict of interest statement
All authors certify that they have no affiliations with or involvement in any organisation or entity with any financial interest (such as honoraria, educational grants; participation in speakers' bureaus; membership, employment, consultancies, stock ownership or other equity interest and expert testimony or patent‐licensing arrangements), or nonfinancial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
The study funders had no role in the study design, collection, interpretation or analysis of the data; report writing, or the decision to submit the paper for publication.
Figures
References
-
- LeResche L., “Epidemiology of Temporomandibular Disorders: Implications for the Investigation of Etiologic Factors,” Critical Reviews in Oral Biology and Medicine 8, no. 3 (1997): 291–305. - PubMed
-
- Schiffman E., Ohrbach R., Truelove E., et al., “Diagnostic Criteria for Temporomandibular Disorders (DC/TMD) for Clinical and Research Applications: Recommendations of the International RDC/TMD Consortium Network* and Orofacial Pain Special Interest Group†,” Journal of Oral & Facial Pain and Headache 28, no. 1 (2014): 6–27. - PMC - PubMed
-
- Lövgren A., Häggman‐Henrikson B., Visscher C. M., Lobbezoo F., Marklund S., and Wänman A., “Temporomandibular Pain and Jaw Dysfunction at Different Ages Covering The Lifespan—A Population Based Study,” European Journal of Pain 20, no. 4 (2016): 532–540. - PubMed
-
- Von Korff M., Dworkin S. F., Le Resche L., and Kruger A., “An Epidemiologic Comparison of Pain Complaints,” Pain 32, no. 2 (1988): 173–183. - PubMed
-
- Manfredini D., Guarda‐Nardini L., Winocur E., Piccotti F., Ahlberg J., and Lobbezoo F., “Research Diagnostic Criteria for Temporomandibular Disorders: A Systematic Review of Axis I Epidemiologic Findings,” Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontics 112, no. 4 (2011): 453–462. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical