Dynamic Kinetic Asymmetric Hydroacylation: Racemization by Soft Enolization
- PMID: 40298317
- PMCID: PMC12082635
- DOI: 10.1021/jacs.5c01753
Dynamic Kinetic Asymmetric Hydroacylation: Racemization by Soft Enolization
Abstract
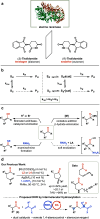
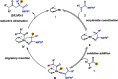
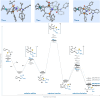
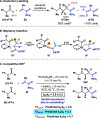
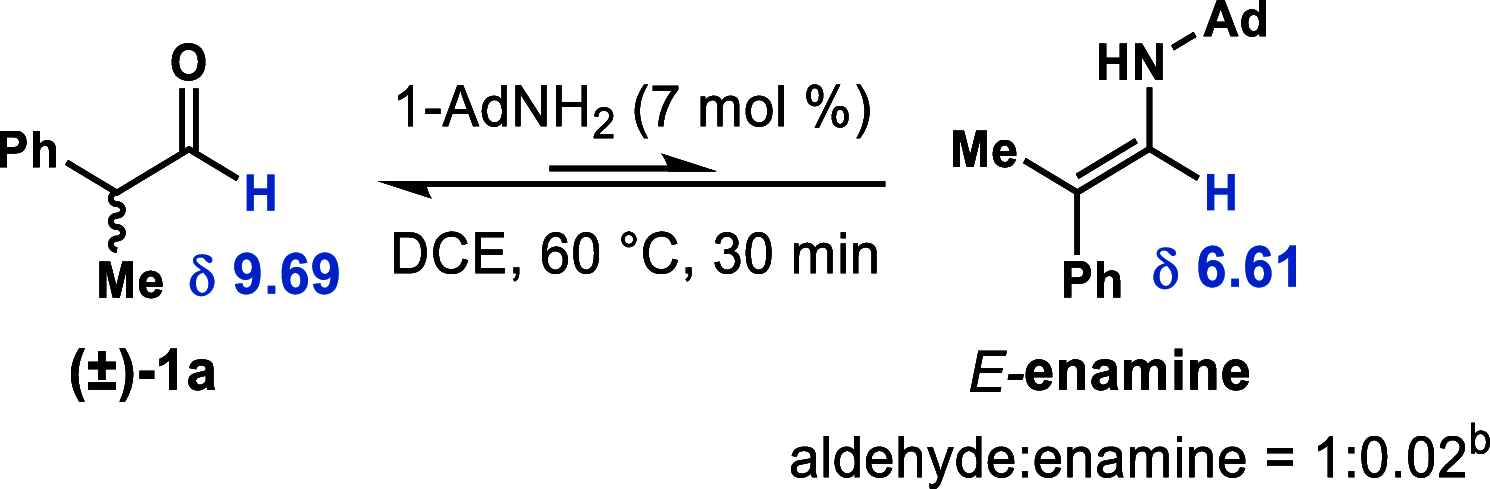
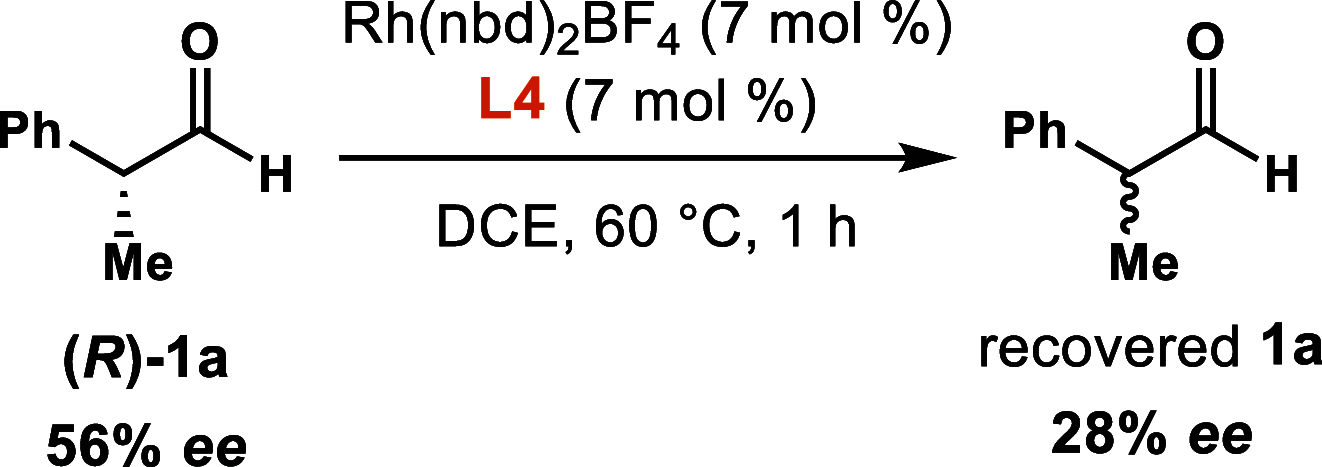
We report a dynamic kinetic asymmetric transformation (DyKAT) of racemic aldehydes by Rh-catalyzed hydroacylation of acrylamides. This intermolecular hydroacylation generates 1,4-ketoamides with high enantio- and diastereoselectivity. DFT and experimental studies provide mechanistic insights and reveal an unexpected Rh-catalyzed pathway for aldehyde racemization. Our study represents a pioneering kinetic resolution by intermolecular hydroacylation and contributes to the growing field of stereoconvergent catalysis featuring C-C bond construction.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Rh(I)-catalyzed intermolecular hydroacylation: enantioselective cross-coupling of aldehydes and ketoamides.J Am Chem Soc. 2014 Jul 2;136(26):9471-6. doi: 10.1021/ja504296x. Epub 2014 Jun 17. J Am Chem Soc. 2014. PMID: 24937681 Free PMC article.
-
Teaching Aldehydes New Tricks Using Rhodium- and Cobalt-Hydride Catalysis.Acc Chem Res. 2021 Mar 2;54(5):1236-1250. doi: 10.1021/acs.accounts.0c00771. Epub 2021 Feb 3. Acc Chem Res. 2021. PMID: 33533586 Free PMC article. Review.
-
Dynamic Kinetic Resolution of Aldehydes by Hydroacylation.Angew Chem Int Ed Engl. 2019 Mar 26;58(14):4705-4709. doi: 10.1002/anie.201900545. Epub 2019 Feb 27. Angew Chem Int Ed Engl. 2019. PMID: 30740841 Free PMC article.
-
Rhodium-catalyzed dynamic kinetic asymmetric transformations of racemic allenes by the [3+2] annulation of aryl ketimines.Angew Chem Int Ed Engl. 2013 Sep 27;52(40):10630-4. doi: 10.1002/anie.201304919. Epub 2013 Aug 16. Angew Chem Int Ed Engl. 2013. PMID: 23956126
-
De-racemization of enantiomers versus de-epimerization of diastereomers--classification of dynamic kinetic asymmetric transformations (DYKAT).Chemistry. 2008;14(27):8060-72. doi: 10.1002/chem.200701643. Chemistry. 2008. PMID: 18512868 Review.
References
-
- Gu S.-X.; Wang H.-F.; Zhu Y.-Y.; Chen F.-E. Natural occurrence, biological functions, and analysis of D-Amino acids. Pharm. Fronts 2020, 02, e79–e8710.1055/s-0040-1713820. - DOI
-
-
For emerging use of epimerization, see:
- Carder H. M.; Wang Y.; Wendlandt A. E. Selective Axial-to-Equatorial epimerization of carbohydrates. J. Am. Chem. Soc. 2022, 144, 11870–11877. 10.1021/jacs.2c04743. - DOI - PMC - PubMed
- Zhang Y.-A.; Palani V.; Seim A. E.; Wang Y.; Wang K. J.; Wendlandt A. E. Stereochemical editing logic powered by the epimerization of unactivated tertiary stereocenters. Science 2022, 378, 383–390. 10.1126/science.add6852. - DOI - PMC - PubMed
- Wang Y.; Carder H. M.; Wendlandt A. E. Synthesis of rare sugar isomers through site-selective epimerization. Nature 2020, 578, 403–408. 10.1038/s41586-020-1937-1. - DOI - PubMed
-
-
-
For a review on stereoconvergent catalysis:
- Bhat V.; Welin E. R.; Guo X.; Stoltz B. M. Advances in Stereoconvergent Catalysis from 2005 to 2015: Transition-Metal-Mediated Stereoablative Reactions, Dynamic Kinetic Resolutions, and Dynamic Kinetic Asymmetric Transformations. Chem. Rev. 2017, 117, 4528–4561. 10.1021/acs.chemrev.6b00731. - DOI - PMC - PubMed
-
Grants and funding
LinkOut - more resources
Full Text Sources

