Cost-Effectiveness Analysis of SGLT2 Inhibitors for Cardio-Renal-Metabolic Disease Based on Data from Japanese Studies
- PMID: 40299278
- PMCID: PMC12085348
- DOI: 10.1007/s12325-025-03157-z
Cost-Effectiveness Analysis of SGLT2 Inhibitors for Cardio-Renal-Metabolic Disease Based on Data from Japanese Studies
Abstract
Introduction: Cardiovascular, renal, and metabolic diseases, collectively known as cardio-renal-metabolic (CRM) disease, interact and exacerbate each other, creating serious clinical and economic burdens. Sodium-glucose cotransporter 2 inhibitors (SGLT2i) are important therapeutic agents in managing CRM disease. Despite proven clinical benefits, the economic benefits of SGLT2i in the management of CRM diseases remain unclear.
Methods: We developed Markov models representing the natural progression of disease for two populations: a type 2 diabetes mellitus (T2DM) population and non-diabetic chronic kidney disease (non-DM CKD) population. These models incorporated key complications, including heart failure, myocardial infarction, stroke, CKD (for the T2DM population), and end-stage renal disease. A systematic literature search was conducted to determine input parameters. For each model, we estimated the 10-year medical costs, quality-adjusted life years (QALY), and incremental cost-effectiveness ratio (ICER) for SGLT2i treatment compared with conventional treatment. A probabilistic sensitivity analysis (PSA) and scenario analyses with conservative assumptions were performed.
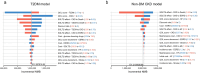
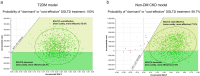
Results: In the base-case analysis, SGLT2i treatment was estimated to increase QALY by 0.177 (7.090 vs 6.913 QALY; T2DM population) and 0.457 (6.980 vs 6.523 QALY; non-DM CKD population), and increase total medical costs by Japanese yen (JPY) 99,060 (JPY 762,524 vs 663,463; T2DM population) and JPY 229,810 (JPY 3,378,873 vs 3,149,063; non-DM CKD population), compared with conventional treatment. The ICER was JPY 559,175/QALY in the T2DM population and JPY 503,123/QALY in the non-DM CKD population. The PSA revealed that the probability of ICER being below the threshold value of JPY 5,000,000/QALY was 100% in the T2DM population and 98.7% in the non-DM CKD population, and the ICERs were below this threshold in all scenario analyses.
Conclusion: SGLT2i treatment was demonstrated to be cost-effective in both the T2DM population and the non-DM CKD population, suggesting the potential of SGLT2i to offer significant clinical and economic benefits in the comprehensive management of CRM diseases.
Keywords: Cardio-renal-metabolic (CRM) diseases; Cardiovascular disease; Chronic kidney disease; Cost-effectiveness; Sodium–glucose cotransporter 2 inhibitors; Type 2 diabetes mellitus.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of Interest: Ataru Igarashi has received lecture fees from Astellas Pharma Inc., Bayer Yakuhin, Ltd., Eli Lilly Japan K.K., Mitsubishi Tanabe Pharma Corporation, MSD Co., Ltd., Novartis Pharma K.K., and Ono Pharmaceutical Co., Ltd.; and belongs to the endowed department sponsored by Takeda Pharmaceutical Co., Ltd. Hisateru Tachimori belongs to the endowed department sponsored by Takeda Pharmaceutical Co., Ltd. Hiroyuki Takagi and Hiroki Akiyama are full-time employees of AstraZeneca K.K. Shun Kohsaka has received research funding from the Japan Society for the Promotion of Science and Pfizer, and has received consulting fees from Novartis and Bristol Myers Squibb, outside of the submitted work. Hiroaki Miyata has received research funding from AstraZeneca K.K. and Mitsubishi Tanabe Pharma Corporation. Keiko Maruyama-Sakurai, Yasumasa Segawa, and Naohiko Imai have no conflicts of interest to declare. Ethical Approval: This study is based on previously conducted studies and published literature and does not contain any new studies with human participants or animals.
Figures


References
-
- Kadowaki T, Komuro I, Morita N, Akiyama H, Kidani Y, Yajima T. Manifestation of heart failure and chronic kidney disease are associated with increased mortality risk in early stages of type 2 diabetes mellitus: analysis of a Japanese real-world hospital claims database. Diabetes Ther. 2022;13:275–86. - PMC - PubMed
-
- Rangaswami J, Bhalla V, Blair JEA, et al. American Heart Association Council on the Kidney in Cardiovascular Disease and Council on Clinical Cardiology. Cardiorenal syndrome: Classification, pathophysiology, diagnosis, and treatment strategies: a scientific statement from the American Heart Association. Circulation. 2019;139:e840–78. - PubMed
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

