TGF-β1 Improves Nerve Regeneration and Functional Recovery After Sciatic Nerve Injury by Alleviating Inflammation
- PMID: 40299436
- PMCID: PMC12024759
- DOI: 10.3390/biomedicines13040872
TGF-β1 Improves Nerve Regeneration and Functional Recovery After Sciatic Nerve Injury by Alleviating Inflammation
Abstract
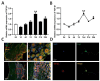
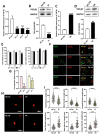
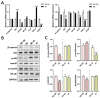
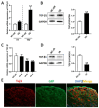
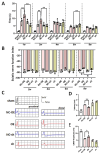
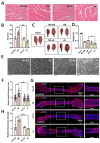
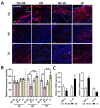
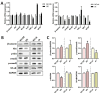
Background: Peripheral nerves have a certain regenerative ability, but their repair and regeneration after injury is a complex process, usually involving a large number of genes and proteins. In a previous study, we analyzed the gene expression profile in rats after sciatic nerve injury and found significant changes in transforming growth factor-beta 1 (TGF-β1) expression, suggesting that TGF-β1 may be involved in the process of nerve regeneration after injury. Methods: In this study, we first detected the time-course expression and localization of TGF-β1 in dorsal root ganglion (DRG) tissues in a rat sciatic nerve transection model via RT-qPCR. Secondly, we investigated the bioactive roles of TGF-β1 in primary cultured DRG neuron cells through a CCK8 assay, TUNEL assay, and immunofluorescence staining. Thirdly, we explored the neuroprotective roles of TGF-β1 in an in vivo model of sciatic nerve regeneration through morphological observation, behavioral, and electrophysiological tests, and a molecular biological measure. Results: We found that TGF-β1 expression was increased after injury and mainly located in the cytoplasm and nuclei of neuron cells in the DRG. TGF-β1 may regulate the viability, apoptosis, and neurite outgrowth of primary DRG neuron cells. In our in vivo model of sciatic nerve regeneration, TGF-β1 improved nerve regeneration and neuronal function recovery after sciatic nerve injury, alleviated the inflammatory response, and relieved neuropathic pain via the TGF-β1/smad2 pathway. Conclusions: This study provides an experimental and theoretical basis for using TGF-β1 as a neuroprotective agent after peripheral nerve injury in clinical practice in the future.
Keywords: TGF-β1; inflammatory response; nerve regeneration; neuronal function recovery; peripheral nerve injury.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Long non-coding RNA uc.217 regulates neurite outgrowth in dorsal root ganglion neurons following peripheral nerve injury.Eur J Neurosci. 2015 Jul;42(1):1718-25. doi: 10.1111/ejn.12966. Epub 2015 Jun 19. Eur J Neurosci. 2015. PMID: 26032672
-
Toll-Like Receptor 4 (TLR4) Promotes DRG Regeneration and Repair after Sciatic Nerve Injury via the ERK-NF-kB Pathway.Mol Neurobiol. 2025 Apr;62(4):4172-4189. doi: 10.1007/s12035-024-04483-z. Epub 2024 Oct 17. Mol Neurobiol. 2025. PMID: 39420131 Free PMC article.
-
Augmented Buyang Huanwu Decoction facilitates axonal regeneration after peripheral nerve transection through the regulation of inflammatory cytokine production.J Ethnopharmacol. 2020 Oct 5;260:113063. doi: 10.1016/j.jep.2020.113063. Epub 2020 Jun 4. J Ethnopharmacol. 2020. PMID: 32505841
-
TGF-β1 is critical for Wallerian degeneration after rat sciatic nerve injury.Neuroscience. 2015 Jan 22;284:759-767. doi: 10.1016/j.neuroscience.2014.10.051. Epub 2014 Nov 4. Neuroscience. 2015. PMID: 25451291
-
Transforming growth factor beta 1, a cytokine with regenerative functions.Neural Regen Res. 2016 Oct;11(10):1549-1552. doi: 10.4103/1673-5374.193223. Neural Regen Res. 2016. PMID: 27904475 Free PMC article. Review.
Cited by
-
Single-cell transcriptomic landscape of sciatic nerve after transection injury.J Neuroinflammation. 2025 Aug 23;22(1):205. doi: 10.1186/s12974-025-03514-3. J Neuroinflammation. 2025. PMID: 40849669 Free PMC article.
References
-
- Carvalho C.R., Reis R.L., Oliveira J.M. Fundamentals and Current Strategies for Peripheral Nerve Repair and Regeneration. Adv. Exp. Med. Biol. 2020;1249:173–201. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

