Magnesium-assisted hydrogen improves isoproterenol-induced heart failure
- PMID: 40300881
- PMCID: PMC12124708
- DOI: 10.4103/mgr.MEDGASRES-D-24-00135
Magnesium-assisted hydrogen improves isoproterenol-induced heart failure
Abstract
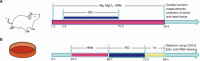
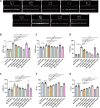
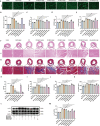
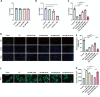
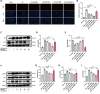
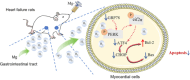
Heart failure (HF) is a leading cause of mortality among patients with cardiovascular disease and is often associated with myocardial apoptosis and endoplasmic reticulum stress (ERS). While hydrogen has demonstrated potential in reducing oxidative stress and ERS, recent evidence suggests that magnesium may aid in hydrogen release within the body, further enhancing these protective effects. This study aimed to investigate the cardioprotective effects of magnesium in reducing apoptosis and ERS through hydrogen release in a rat model of isoproterenol (ISO)-induced HF. Magnesium was administered orally to ISO-induced HF rats, which improved cardiac function, reduced myocardial fibrosis and cardiac hypertrophy, and lowered the plasma levels of creatine kinase-MB, cardiac troponin-I, and N-terminal B-type natriuretic peptide precursor in ISO-induced HF rats. It also inhibited cardiomyocyte apoptosis by upregulating B-cell lymphoma-2, downregulating Bcl-2-associated X protein, and suppressing ERS markers (glucose-related protein 78, activating transcription factor 4, and C/EBP-homologous protein). Magnesium also elevated hydrogen levels in blood, plasma, and cardiac tissue, as well as in artificial gastric juice and pure water, where hydrogen release lasted for at least four hours. Additionally, complementary in vitro experiments were conducted using H9C2 cardiomyocyte injury models, with hydrogen-rich culture medium as the intervention. Hydrogen-rich culture medium improved the survival and proliferation of ISO-treated H9C2 cells, reduced the cell surface area, inhibited apoptosis, and downregulated ERS pathway proteins. However, the protective effects of hydrogen were negated by tunicamycin (an inducer of ERS) in H9C2 cells. In conclusion, magnesium exerts significant cardioprotection by mitigating ERS and apoptosis through hydrogen release effects in ISO-induced HF.
Keywords: H9C2 cells; apoptosis; cardiac hypertrophy; endoplasmic reticulum stress; heart failure; hydrogen; isoproterenol; magnesium; myocardial fibrosis; rats.
Copyright © 2025 Medical Gas Research.
Conflict of interest statement
Figures


Similar articles
-
Chlorogenic acid attenuates cardiac hypertrophy via up-regulating Sphingosine-1-phosphate receptor1 to inhibit endoplasmic reticulum stress.ESC Heart Fail. 2024 Jun;11(3):1580-1593. doi: 10.1002/ehf2.14707. Epub 2024 Feb 19. ESC Heart Fail. 2024. PMID: 38369950 Free PMC article.
-
Isoproterenol instigates cardiomyocyte apoptosis and heart failure via AMPK inactivation-mediated endoplasmic reticulum stress.Apoptosis. 2013 Jul;18(7):800-10. doi: 10.1007/s10495-013-0843-5. Apoptosis. 2013. PMID: 23620435
-
[Effects of total flavonoids of Dracocephalum moldavica on apoptosis of H9c2 cells induced by OGD/R injury and endoplasmic reticulum stress].Zhongguo Zhong Yao Za Zhi. 2025 Mar;50(5):1321-1330. doi: 10.19540/j.cnki.cjcmm.20241212.703. Zhongguo Zhong Yao Za Zhi. 2025. PMID: 40350915 Chinese.
-
Shikonin ameliorates isoproterenol (ISO)-induced myocardial damage through suppressing fibrosis, inflammation, apoptosis and ER stress.Biomed Pharmacother. 2017 Sep;93:1343-1357. doi: 10.1016/j.biopha.2017.06.086. Epub 2017 Jul 24. Biomed Pharmacother. 2017. PMID: 28753907
-
Effects of the Activin A-Follistatin System on Myocardial Cell Apoptosis through the Endoplasmic Reticulum Stress Pathway in Heart Failure.Int J Mol Sci. 2017 Feb 10;18(2):374. doi: 10.3390/ijms18020374. Int J Mol Sci. 2017. PMID: 28208629 Free PMC article.
References
-
- Savarese G, Becher PM, Lund LH, Seferovic P, Rosano GMC, Coats AJS. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovasc Res. 2023;118:3272–3287. - PubMed
-
- Cai Q, Li Y, Zhang Y, et al. Xinshubao tablet ameliorates myocardial injury against heart failure via the DCN/PPARα/PGC-1α/P300 pathway. Biomed Pharmacother. 2023;166:115285. - PubMed
-
- Wang Q, Liang S, Qian J, et al. OTUD1 promotes isoprenaline- and myocardial infarction-induced heart failure by targeting PDE5A in cardiomyocytes. Biochim Biophys Acta Mol Basis Dis. 2024;1870:167018. - PubMed
-
- Chouairi F, Levin A, Biegus J, Fudim M. Emerging devices for heart failure management. Prog Cardiovasc Dis. 2024;82:125–134. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

