Spatial-temporal interactions between white matter hyperintensities and multiple pathologies across the Alzheimer's disease continuum
- PMID: 40302045
- PMCID: PMC12040729
- DOI: 10.1002/alz.70098
Spatial-temporal interactions between white matter hyperintensities and multiple pathologies across the Alzheimer's disease continuum
Abstract
Introduction: The interactive relationships between Alzheimer's disease (AD) and white matter hyperintensities (WMHs) in multiscale brain structural networks still need to be clarified.
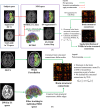
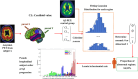
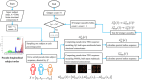
Methods: Based on subjects enrolled from the Alzheimer's Disease Neuroimaging Initiative (ADNI) database, regional WMHs, amyloid beta (Aβ) accumulation, and microstructural changes detected by diffusion weighted imaging (DWI) in multiscale brain networks were modeled by time-evolving graphs; their interactive relationships were further investigated using Granger causality after constructing pseudo-time subject sequences.
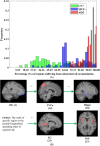
Results: In up to 86% of the extracted pseudo-time subject sequences, Aβ was determined to be the Granger cause of WMHs in the structural connectivity of the inferior longitudinal fasciculus (ILF). Meanwhile WMHs were significantly correlated with microstructural changes measured by reduced fractional anisotropy in the inferior fronto-occipital fasciculus, ILF, and cingulum, which Granger causality pathways detected in 91%, 94%, and 93% of pseudo-time subject sequences, respectively.
Discussion: These findings provide novel insights for understanding the multiscale space-time interactions between WMHs and AD pathologies.
Highlights: This study proposed time-evolving graph modeling of heterogeneous disease markers (amyloid beta [Aβ], white matter hyperintensities [WMHs], and microstructural changes of white matter tracts) across the Alzheimer's disease (AD) continuum to investigate their complex interactions in multiscale brain structural networks. Regional accumulation of Aβ promoted WMH progression in subnetworks connected by the inferior longitudinal fasciculus (ILF). Regional WMHs were strongly associated with bundle-specific microstructural changes in the ILF, inferior fronto-occipital fasciculus, and cingulum. These results might provide novel insights for understanding the interactive relationship between cerebral small vessel disease and AD.
Keywords: Alzheimer's disease; brain structural connectomes; causal inference; time‐evolving graph; white matter hyperintensities.
© 2025 The Author(s). Alzheimer's & Dementia published by Wiley Periodicals LLC on behalf of Alzheimer's Association.
Conflict of interest statement
The authors declare no competing interest. Author disclosures are available in the supporting information.
Figures
References
-
- Garnier‐Crussard A, Cotton F, Krolak‐Salmon P, Chételat G. White matter hyperintensities in Alzheimer's disease: beyond vascular contribution. Alzheimers Dement. 2023;19(8):3738‐3748. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

