Taxus chinensis-Derived Nanovesicles Alleviate Mouse Colitis by Inhibiting Inflammatory Cytokines and Restoring Gut Microbiota
- PMID: 40303007
- PMCID: PMC12039931
- DOI: 10.2147/JIR.S513823
Taxus chinensis-Derived Nanovesicles Alleviate Mouse Colitis by Inhibiting Inflammatory Cytokines and Restoring Gut Microbiota
Abstract
Background: Recent research has increasingly focused on plant-derived products as potential alternatives to chemotherapeutic agents, aiming to reduce side effects. Among these, plant-derived nanovesicles (NVs) have garnered significant attention for their potential in treating colitis.
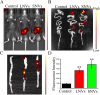
Methods: In this study, we extracted NVs from the leaves (LNVs) and stems (SNVs) of Taxus, a well-known natural anti-cancer plant. The targeting ability of these NVs was evaluated in the mouse colon using an IVIS imaging system. Additionally, we assessed the therapeutic effects of these plant-derived NVs on ulcerative colitis in a mouse model.
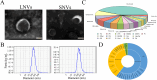
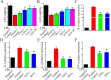
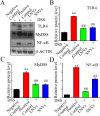
Results: Our findings reveal that the NVs exhibit an ideal vesicle size of 150.0 nm and contain a rich array of lipids, functional proteins, and bioactive small molecules. In vitro anti-inflammatory experiments demonstrated that both LNVs and SNVs enhanced cell viability and reduced levels of pro-inflammatory cytokines. Importantly, neither LNVs nor SNVs induced significant cytotoxicity. In vivo, oral administration of LNVs and SNVs ameliorated colitis-related symptoms in mice and accelerated colitis resolution by suppressing the TLR4/MyD88/NF-κB pathway and reducing levels of pro-inflammatory cytokines, including IL-1β, IL-6, and TNF-α. Furthermore, 16S rDNA sequencing data suggested that LNVs play a crucial role in regulating gut microbiota.
Conclusion: Collectively, our findings suggest that plant-derived NVs from Taxus represent a promising novel natural nanomedicine for use as an anti-inflammatory agent in the treatment of colonic diseases.
Keywords: colitis; gut microbiota; plant-derived nanovesicles.
© 2025 Gong et al.
Conflict of interest statement
The authors declare no conflicts of interest in this work.
Figures



Similar articles
-
Garlic-derived exosome-like nanovesicles alleviate dextran sulphate sodium-induced mouse colitis via the TLR4/MyD88/NF-κB pathway and gut microbiota modulation.Food Funct. 2023 Aug 14;14(16):7520-7534. doi: 10.1039/d3fo01094e. Food Funct. 2023. PMID: 37523213
-
Turmeric-derived nanovesicles as novel nanobiologics for targeted therapy of ulcerative colitis.Theranostics. 2022 Jul 18;12(12):5596-5614. doi: 10.7150/thno.73650. eCollection 2022. Theranostics. 2022. PMID: 35910802 Free PMC article.
-
Kaempferol Alleviates Murine Experimental Colitis by Restoring Gut Microbiota and Inhibiting the LPS-TLR4-NF-κB Axis.Front Immunol. 2021 Jul 22;12:679897. doi: 10.3389/fimmu.2021.679897. eCollection 2021. Front Immunol. 2021. PMID: 34367139 Free PMC article.
-
Hemp-Derived Nanovesicles Protect Leaky Gut and Liver Injury in Dextran Sodium Sulfate-Induced Colitis.Int J Mol Sci. 2022 Sep 1;23(17):9955. doi: 10.3390/ijms23179955. Int J Mol Sci. 2022. PMID: 36077356 Free PMC article.
-
Milk-derived extracellular vesicles alleviate ulcerative colitis by regulating the gut immunity and reshaping the gut microbiota.Theranostics. 2021 Jul 25;11(17):8570-8586. doi: 10.7150/thno.62046. eCollection 2021. Theranostics. 2021. PMID: 34373759 Free PMC article.
References
LinkOut - more resources
Full Text Sources

