Mechanical stress contributes to ligamentum flavum hypertrophy by inducing local inflammation and myofibroblast transition in the innovative surgical rabbit model
- PMID: 40303410
- PMCID: PMC12037363
- DOI: 10.3389/fimmu.2025.1541577
Mechanical stress contributes to ligamentum flavum hypertrophy by inducing local inflammation and myofibroblast transition in the innovative surgical rabbit model
Abstract
Background: Lumbar spinal canal stenosis (LSCS) ranks as a prevalent spinal disorder in senior populations. Ligamentum flavum hypertrophy (LFH) is a significant feature of LSCS, yet its cause is unclear. The purpose of this study was to create a novel animal model for LFH and explore the pathological mechanisms involved.
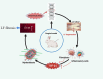
Methods: A novel rabbit model for intervertebral mechanical stress concentration was established through posterolateral fusion using steel wire. Radiological analysis and biological validation were used to determine the crucial role of mechanical stress in LFH and explore the effect of this animal model.
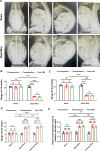
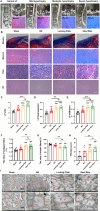
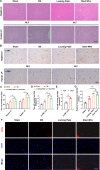
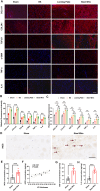
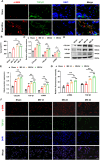
Results: After 12 weeks, the LF subjected to mechanical stress concentration exhibited a disruption and reduction in elastic fibers, collagen accumulation, increased thickness of LF, elevated LF cells, and increased levels of certain factors related to fibrosis and inflammation. These findings were histologically consistent to those found in human LFH. Furthermore, in vitro, mechanical stretch was discovered to enhance the conversion of fibroblasts into myofibroblasts by boosting TGF-β1 secretion in LF fibroblasts. In addition, compared to conventional internal fixation, this new surgical model provided advantages such as minor damage, decreased bleeding, and reduced technical difficulty and molding costs.
Conclusion: This novel rabbit model is able to replicate the moderate pathological features of human LFH. Mechanical stress is an independent factor leading to LFH, which can promote the TGF-β1 secretion in LF cells and some inflammatory cells, subsequently induce the myofibroblast transition, and finally result in collagen accumulation and LF fibrosis.
Keywords: TGF-β1; fibrosis; ligamentum flavum; mechanical stress; rabbit model.
Copyright © 2025 Ma, Feng, Chen, Zhang and Sun.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

