Ultrasound-assisted synthesis of biomass-derived carbon-supported binary metal oxides for efficient adsorption of heavy metals from wastewater
- PMID: 40303560
- PMCID: PMC12038807
- DOI: 10.1039/d5ra00057b
Ultrasound-assisted synthesis of biomass-derived carbon-supported binary metal oxides for efficient adsorption of heavy metals from wastewater
Abstract
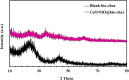
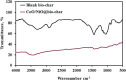
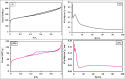
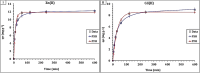
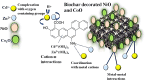
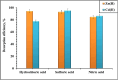
Heavy metal contamination in water sources remains a critical environmental issue, primarily due to industrial activities, in terms of its continuous contribution to pollution through non-compliance and illegal discharge. This study presents an innovative biochar-supported binary metal oxide composite (nickel oxide (NiO) and cobalt oxide (CoO) nanoparticles) synthesized via ultrasound-assisted techniques for efficient adsorption of Zn(ii) and Cd(ii) ions from wastewater. By utilizing solid residues and leveraging ultrasound technology, this approach aligns with the principles of green chemistry due to utilization of a renewable biomass-based source, enhancing energy efficiency in the synthesis process, and minimizing waste production. Thereby a sustainable and innovative route for material development is explicitly demonstrated. Structural and morphological characterizations confirm the uniform integration of Nickel oxide (NiO) and cobalt oxide (CoO) particles into the biochar matrix, leading to maximum adsorption capacities of 18.9 mg g-1 for Zn(ii) and 10.2 mg g-1 for Cd(ii). The adsorption process follows a chemisorptive monolayer mechanism, as demonstrated by kinetic and isotherm studies, and is thermodynamically confirmed to be endothermic and spontaneous. The material also exhibits excellent reusability over five adsorption-desorption cycles. By integrating sustainable resources with innovative synthesis techniques, this work contributes to advancing wastewater remediation technologies while supporting global sustainability initiatives.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







References
-
- Gupta V. K. Nayak A. Bhushan B. Agarwal S. Crit. Rev. Environ. Sci. Technol. 2015;45:613–668. doi: 10.1080/10643389.2013.876526. - DOI
-
- Gupta V. K. Ganjali M. R. Nayak A. Bhushan B. Agarwal S. Chem. Eng. J. 2012;197:330–342. doi: 10.1016/j.cej.2012.04.104. - DOI
-
- El-Said A. G. Badawy N. A. Garamon S. E. Environ. Anal. Toxicol. 2018;8:1–5.
-
- Nayak A. Bhushan B. Gupta V. Rodriguez-Turienzo L. J. Environ. Chem. Eng. 2016;4:4342–4353. doi: 10.1016/j.jece.2016.10.001. - DOI
LinkOut - more resources
Full Text Sources

